|
简介
慢病毒载体(Lentiviral vectors, LVs)是在人免疫缺陷病毒(HIV-1病毒)基础上改造而成的病毒载体系统,它能高效的将目的基因(或RNAi)导入动物和人的原代细胞或细胞系,对分裂细胞和非分裂细胞均具有感染能力。
慢病毒包装系统
慢病毒包装系统主要有慢病毒表达载体和包装载体及胞膜载体构成,一般由三个(二代系统)或者四个质粒(三代系统)构成,包装病毒所用细胞一般为293系列的细胞,其主要过程是将组成慢病毒系统的质粒共转染293细胞,48-72h后收集细胞培养基上清,经过浓缩纯化后的到慢病毒粒子。
载体优势
1.对分裂期细胞和非分裂期细胞均有感染作用2.操作方便,安全性高 3.表达稳定高效 4.实验周期短 5.多种载体可选
载体应用
1.将目的基因/RNAi基因转入难以转染的细胞,比如神经元细胞、干细胞或其它原代细胞。2.将目的基因/RNAi基因转入动物组织,以期获得长期表达。 3.构建稳定表达目的蛋白/RNAi的细胞系,再用ex vivo的方法导入动物体内。 4.基因治疗 5.转基因动物 6.基因敲除 7.药物研究:构建表达受体蛋白的细胞系,研究药物的作用。 8.快速建立生产目的蛋白的细胞系,非常有前途的真核细胞表达方法。
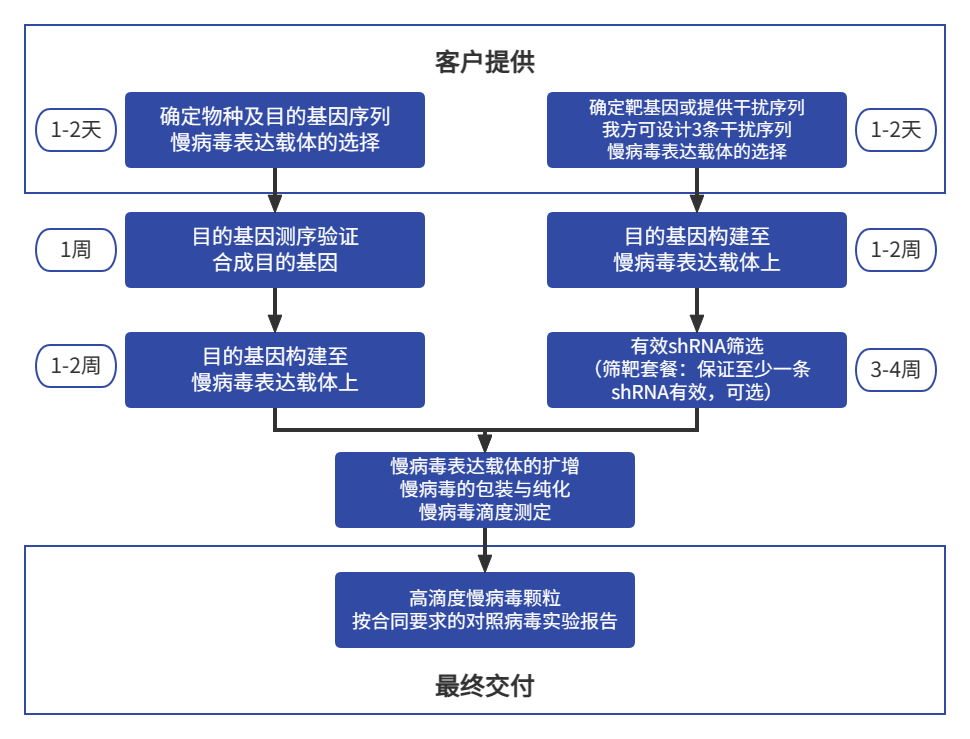
服务说明
案例展示
一、慢病毒载体在神经科学领域的应用
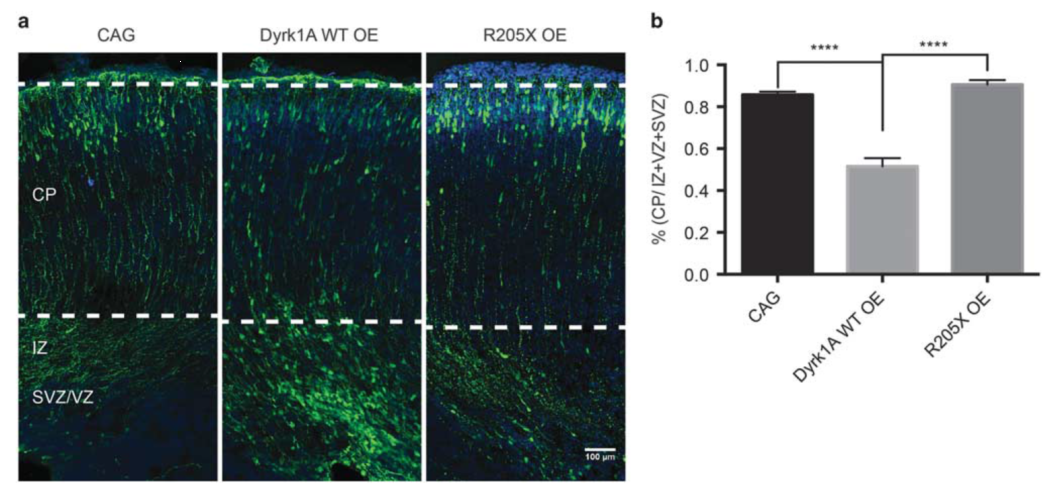
慢病毒搭载shRNA后干扰小鼠杏仁核内Akap5的表达,下调小鼠脑内BLA中Akap5的含量,可有效缓解小鼠出现慢性束缚造成的焦虑/抑郁样行为。(Zhou et al., Biol Psychiatry. 2019) 
2.慢病毒标记CNS细胞
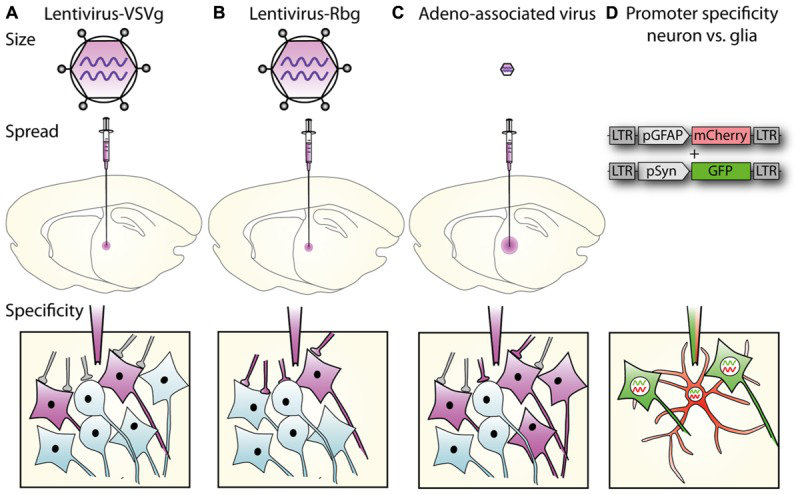
3.钙成像
4.慢病毒(LV)结合狂犬病毒(RV)操控活性依赖的神经元
5.慢病毒介导基因沉默研究中间神经元电偶联
6.慢病毒在光遗传学中的应用

7.慢病毒的药理学应用
二、慢病毒载体在其他领域的应用
2. 慢病毒介导的造血干细胞治疗X连锁严重免疫缺陷疾病(SCID-X1)接受SIN-LV治疗后,SCID-X1病人T细胞和B细胞的功能改善。密码子优化的 Self-inactivating element LV,即SIN-LV(Cl20-i4-Ef1a-hγcOPT) 转导CD34+HSC后回输给病人。上图显示病人的CD3T 细胞能够响应药物刺激增殖(A),治疗后的12-16周,病人出现 T1B cells(CD10++CD21lo)向T2/3 B cells+hi (CD10++CD21lo)的转变,免疫细胞类群构成多样性增加。(De Ravin et al.,Sci Transl Med, 2016)
3. LV-CRISPR/Cas9清除人T细胞基因组中的HIV-1
4. LV-CRISPRa/i介导转录激活和抑制
1. Dang T, Duan W Y, Yu B, et al. Autism-associated Dyrk1a truncation mutants impair neuronal dendritic and spine growth and interfere with postnatal cortical development[J]. Molecular Psychiatry, 2017. 2. Cetin, A. and E.M. Callaway, Optical control of retrogradely infected neurons using drug-regulated "TLoop" lentiviral vectors. J Neurophysiol, 2014. 111(10): p. 2150-9. 3. Wei, Y., et al., Lentiviral vectors enveloped with rabies virus glycoprotein can be used as a novel retrograde tracer to assess nerve recovery in rat sciatic nerve injury models. Cell Tissue Res, 2014. 355(2): p. 255-66. 4. Parr-Brownlie, L.C., et al., Lentiviral vectors as tools to understand central nervous system biology in mammalian model organisms. Front Mol Neurosci, 2015. 8: p. 14. 5. Parr-Brownlie, Louise C., et al. "Lentiviral vectors as tools to understand central nervous system biology in mammalian model organisms."Frontiers in Molecular Neuroscience.8(2015). 6. De Juan-Sanz J , Holt G T , Schreiter E R , et al. Axonal Endoplasmic Reticulum Ca 2+, Content Controls Release Probability in CNS Nerve Terminals[J]. Neuron, 2017, 93(4):867-881.e6. 7. Sakurai K , Zhao S , Takatoh J , et al. Capturing and Manipulating Activated Neuronal Ensembles with CANE Delineates a Hypothalamic Social-Fear Circuit[J]. Neuron, 2016:S0896627316307164. 8. Yao X H , Wang M , He X N , et al. Electrical coupling regulates layer 1 interneuron microcircuit formation in the neocortex[J]. Nature Communications, 2016, 7:12229. 9. Zou D , Chen L , Deng D , et al. DREADD in Parvalbumin Interneurons of the Dentate Gyrus Modulates Anxiety, Social Interaction and Memory Extinction[J]. Current Molecular Medicine, 2016, 16(1):91-102. 10. Chen B , Gilbert L , Cimini B , et al. Dynamic Imaging of Genomic Loci in Living Human Cells by an Optimized CRISPR/Cas System[J]. Cell, 2013, 155(7):1479-1491. 11. De Ravin S S , Wu X , Moir S , et al. Lentiviral hematopoietic stem cell gene therapy for X-linked severe combined immunodeficiency[J]. Science Translational Medicine, 2016, 8(335):335ra57-335ra57. 12. Kaminski R , Chen Y , Fischer T , et al. Elimination of HIV-1 Genomes from Human T-lymphoid Cells by CRISPR/Cas9 Gene Editing[J]. Scientific Reports, 2016, 6:22555. 13. Genome-Scale CRISPR-Mediated Control of Gene Repression and Activation[J]. Cell, 2014, 159(3):647-661. 14. Zhou, H. Y., J. G. He, Z. L. Hu, S. G. Xue, J. F. Xu, Q. Q. Cui, S. Q. Gao, B. Zhou, P. F. Wu, L. H. Long, F. Wang and J. G. Chen 2019. A-Kinase Anchoring Protein 150 and Protein Kinase A Complex in the Basolateral Amygdala Contributes to Depressive-like Behaviors Induced by Chronic Restraint Stress. Biol Psychiatry 86: 131-142. doi: 10.1016/j.biopsych.2019.03.967 如有相关需求,或了解更多产品服务,欢迎咨询我们!
|