Nonradioactive Methods for Detecting Activation of Ras-Related Small G Proteins
互联网
互联网
相关产品推荐

Estrogen Related Receptor gamma Rabbit pAb(bs-6213R)-50ul/100ul/200ul
¥1180

HB Western blotting Principles and Methods
¥223

Recombinant-Drosophila-melanogaster-CAAX-prenyl-protease-2SrasCAAX prenyl protease 2 EC= 3.4.22.- Alternative name(s): Farnesylated proteins-converting enzyme 2; FACE-2 Prenyl protein-specific endoprotease 2 Protein severas
¥11466

HUMAN EGF DETECTING MONOCLONAL-M807
¥4586
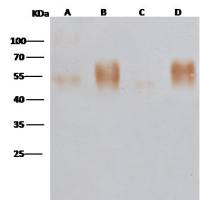
K-Ras 兔多抗 (抗原亲和纯化)
¥600
相关问答

