相关产品推荐更多 >
万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 询价记录
- 文献和实验
- 技术资料
- 英文名:
ReadiPrep™ Nuclear/Cytoplasmic Fractionation Kit
- 供应商:
上海研卉生物科技有限公司
- 库存:
4W
- 规格:
50T
ReadiPrep™ Nuclear/Cytoplasmic Fractionation Kit
细胞核质蛋白分离试剂盒促销/细胞质蛋白分离试剂盒价格/细胞核蛋白分离试剂盒优惠价。
ReadiPrep™核/细胞质分离试剂盒提供了从哺乳动物细胞或组织中逐步分离核和细胞质提取物的方法。分离出的蛋白质具有高浓度,并保持其生物活性。ReadiPrep™核/细胞质分离试剂盒的核和细胞质提取物与许多下游应用兼容,包括酶活性测定和荧光蛋白印迹等。
产品说明书
样品实验方案
简要概述
- 用PBS冲洗细胞
- 加入500 µL细胞质提取缓冲液
- 离心20秒
- 收集上清液(细胞质提取物)
- 将沉淀重新悬浮在150 µL 1X高盐缓冲液中
- 离心20分钟
- 收集上清液(核提取物)
溶液配制
工作溶液配制
1.细胞液提取缓冲液(1X):将100 µL的10X蛋白酶抑制剂(组分C)和1 µL的DTT(组分D)添加到0.9 mL的细胞质提取缓冲液(组分A)中。
2.核提取缓冲液(1X):将100 µL 10X蛋白酶抑制剂(组分C)和1 µL DTT(组分D)添加到0.9 mL核提取缓冲液(组分B)中。
注意:0.5 mL 1X细胞质提取缓冲液和150 µL 1X核提取缓冲液足以进行1次测定,并根据需要准备新鲜的缓冲液。
实验步骤
1.用冷PBS冲洗细胞1次。 对于悬浮的细胞,请通过离心收集细胞。
2.向细胞中加入500 µL 1X Cytosol提取缓冲液。 对于贴壁细胞,将贴壁细胞刮入1.5 mL离心管中。
3.剧烈涡旋以完全重悬细胞。
4.以16,000 g离心1-2分钟,然后将上清液(细胞质提取物)转移到另一个干净的试管中。 将试管放在冰上以备下游使用或在-80°C下保存。
5.将沉淀重悬于150 µL 1X核提取缓冲液中。
6.剧烈涡旋以完全重悬沉淀。
7.将试管在4°C旋转30分钟。
8.以16,000g离心20分钟,然后将上清液(核提取物)转移到另一个干净的试管中。 将试管放在冰上以备下游使用或在-80°C下保存。
图示

图1.使用ReadiPrep 核/细胞质分离试剂盒(AATBioquest®,Cat#60000)收集Hela细胞的核和细胞质提取物。 通过Amplite 荧光荧光胺蛋白质定量试剂盒(AATBioquest®,Cat#11100)对蛋白质进行定量。 A.在有或没有HDAC抑制剂曲古抑菌素A的情况下孵育8ug核或细胞质提取物,并通过Amplite 荧光HDAC活性测定试剂盒(AATBioquest®,Cat#13601)测量HDAC活性。 B.使用40ug总蛋白的核或细胞质提取物。 2 ug / ml兔抗HDAC1抗体用于探测硝酸纤维素膜过夜。 使用了10 ug / ml的iFluor 647山羊抗兔IgG(H + L)(AATBioquest®,Cat#16809)。 该检测通过UVP多光谱成像系统(Biolite)可视化。 M:标记,Nu:核提取物,细胞:细胞质提取物。 |
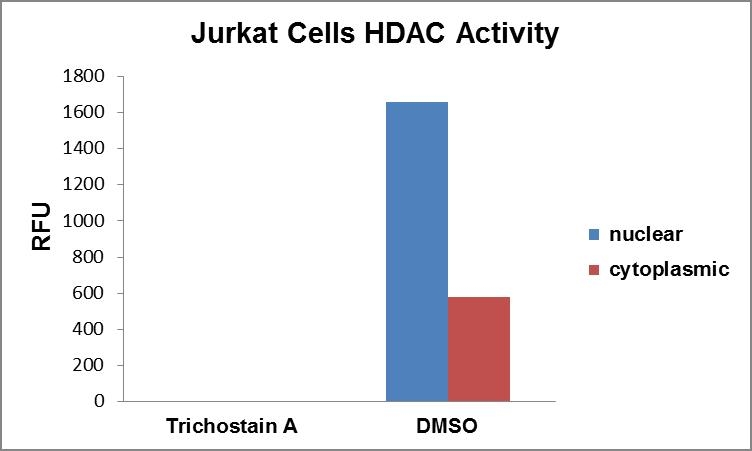
图2.使用ReadiPrep Nucleus / Cytosol分离试剂盒收集了Jurkat细胞的核和细胞质提取物。 通过Amplite 荧光荧光胺蛋白质定量试剂盒(AATBioquest®,Cat#11100)对蛋白质进行定量。 在有或没有HDAC抑制剂Trichostain A的情况下孵育8 µg核或细胞质提取物,并通过Amplite 荧光HDAC活性测定试剂盒(AATBioquest®,Cat#13601)测量HDAC活性。 |

图3.使用ReadiPrep Nucleus / Cytosol Fractionation Kit收集了Hela细胞的核和细胞质提取物。 |
Components
| Component A: Cytosol Extraction Buffer | 1 Bottle (25 mL) |
| Component B: Nuclear Extraction Buffer | 1 Bottle (8 mL) |
| Component C: 10X Protease Inhibitors | 1 Vial (4 mL) |
| Component D: DTT (1 M) | 1 Vial (100 uL) |
Example protocol
At a glance
Protocol summary
- Rinse cells with PBS
- Add 500 µL of Cytosol Extraction Buffer
- Centrifuge for 20 seconds
- Collect the supernatant (Cytoplasmic extract)
- Re-suspend the pellets in 150 µL 1X High Salt Buffer
- Centrifuge for 20 minutes
- Collect the supernatant (Nuclear Extract)
Important notes
Thaw all the kit components at room temperature before starting the experiment (you may store buffers at 4°C or –20°C), keep the buffers on ice during the experiment.
Preparation of working solution
1. Cytosol Extraction Buffer (1X):
Add 100 µL of 10X Protease Inhibitors (Component C) and 1 µL of DTT (Component D) into 0.9 mL of Cytosol Extraction Buffer (Component A).
2. Nuclear Extraction Buffer (1X):
Add 100 µL of 10X Protease Inhibitors (Component C) and 1 µL of DTT (Component D) into 0.9 mL of Nuclear Extraction Buffer (Component B).
Note: 0.5 mL 1X Cytosol Extraction Buffer and 150 µL of 1X Nuclear Extraction Buffer is enough for 1 assay, prepare fresh buffer as needed.
Procedure
- Rinse the cells 1 time with cold PBS. For cells in suspension, collect the cells by centrifugation.
- Add 500 µL of 1X Cytosol Extraction Buffer to cells. For adherent cells, scrape the adherent cells into a 1.5 mL centrifuge tube.
- Vortex vigorously to fully re-suspend the cells.
- Centrifuge at 16,000 g for 1 - 2 minutes and transfer the supernatant (Cytoplasmic extract) to another clean tube. Keep the tube on ice for downstream applications or store at -80°C.
- Re-suspend the pellet in 150 µL 1X Nuclear Extraction Buffer.
- Vortex vigorously to fully re-suspend the pellet.
- Rotate the tube at 4°C for 30 minutes.
- Centrifuge at 16,000g for 20 minutes and transfer the supernatant (Nuclear extract) to another clean tube. Keep the tube on ice for downstream applications or store at -80°C.
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
- 作者
- 内容
- 询问日期
 文献和实验
文献和实验:总膜,细胞核,细胞质,细胞器及质膜。同样起始样品量,只需设定离心力和离心时间,结果的一致性更好,实验间的差异可达最小化。与溶液法试剂盒相比,基于离心管柱法的细胞分离试剂盒更加简便,强大,可获得交叉污染很少的亚细胞结构。离心管柱法细胞组分分离试剂盒在全世界范围获得一致好评。
。 图 1. 富集质膜组分后检测跨膜蛋白更易检出:目的蛋白 SGL1 是跨膜蛋白,使用总膜蛋白检测困难,使用柱式法质膜及细胞组分分离试剂盒富集质膜后再检测质膜组分,可以显著提高 SGL1 的检出效率。 (图片来源:Kashiwagi Y,2015,PLoS ONE 10(6):e0130605. doi:10.1371/journal.pone.0130605.) (3)有条件诱导目的蛋白表达 如果通过增溶目的蛋白或分离亚细胞结构富集目的蛋白,仍无法得到 WB 条带,可通过查阅相关
等渗的提取缓冲液(带或不带还原剂和蛋白酶抑制剂)来维持感兴趣的细胞器。有时也需要机械裂解或去污剂裂解。“各个不同供应商在细胞分级分离试剂盒的设计上都是一致的,而不同细胞组分的差异在于缓冲液组成、裂解试剂和离心速度,”Moehlenbrock说。 此类试剂盒的价值并不在于某些秘密试剂,而在于QC系统的一致性和重复性。“细胞分级分离的方法已经过充分鉴定,其操作步骤适用于大部分应用,”Moehlenbrock说。“商业化试剂盒为最终用户带来了简便和效率,同时提供了品质保证的安全
 技术资料
技术资料暂无技术资料 索取技术资料