万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 文献和实验
- 技术资料
- 库存:
大量
- 供应商:
北京百奥创新科技有限公司
- 规格:
5mL;50mL;1L
NHS活化磁性琼脂糖介质4HF
产品名称:NHS活化磁性琼脂糖介质4HF英文名称:NHS-activated SepFast MAG 4HF
Product Introduction:
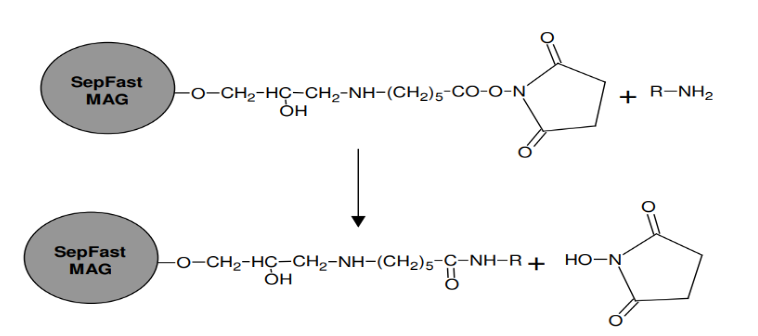
NHS (N-hydroxysuccinimide)-activated SepFast MAG is used to make magnetic adsorbents coupled with molecules containing amine groups. Coupling biospecific ligands to NHS-activated agarose is a successful and well-documented technique. The coupling reaction is spontaneous, rapid and easy to carry out. No toxic chemicals or special equipment is required.
NHS-activated SepFast MAG media forms chemically stable amide bonds with ligands containing primary amine groups. The activated NHS group has a long spacer arm (11 atoms) that is particularly helpful in the immobilization of structurally restricted ligands such as proteins and peptides.
This pre-activated resin can be readily employed to make various magnetic affinity chromatography adsorbents for both small scale and large scale purification applications. After binding of target molecules, the magnetic resin can be readily isolated from the biological feedstocks with the aid of a magnet.
Product Properties:
NHS activated SepFast MAG 4HF is made of highly cross-linked 4% agarose containing magnetic material. It shows high mechanical rigidity and allows harsher operational conditions.
Agarose has long been used for chromatographic separations due to its excellent hydrophilic and low non-specific-binding nature. The particles have an open pore structure with excellent mass transfer properties to various molecules.
The base matrix is activated through a long hydrophilic spacer arm (11 atoms) with Nhydroxysuccinimide at the end. It reacts directly with the primary amine groups in molecules to be immobilized to form stable amide bonds. NHS-activated SepFasm MAG media is supplied as a suspension in 100% isopropanol.

Product Series:

风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 文献和实验
文献和实验S.L. Lim, H.W. Ng, M.A. Akwiditya, C.W. Ooi, E.S. Chan, K.L. Ho, W.S. Tan, G.K. Chua, B.T. Tey, Single-step purification of recombinant hepatitis B core antigen Y132A dimer from clarified Escherichia coli feedstock using a packed bed anion exchange chromatography, Process Biochemistry, 2018, 69:208-215.
后,将EP管置于磁性分离架上,去除上清,加入封闭液,此时,磁珠的终浓度为5mg/mL,同样置于37℃环境旋转混匀反应1h。 2.5. 封闭完成后,重复2.2步骤,最后将磁珠用保存液保存,此时磁珠的浓度可自定。 三、羧基磁性微球偶联抗体 1. 偶联条件 1.1. 活化剂:EDC和NHS 1.2. 反应温度:37℃环境 1.3. 反应时间: 1.3.1.活化时间:30min 1.3.2.偶联时间:2h
作用。已有报道,在可见光(需加光感剂如亚甲基蓝)和紫外光(无需光感剂)条件下核酸可与蛋白质直接交联。反应膜 活化膜的制造大受关注。所谓活化膜是指表面可提供直接反应的化学基团的膜介质。无需其它试剂,液体样本可直接结合到活化膜表面,这样体现它的好多优势,特别是应用到微量分析中。用来活化膜介质的化学试剂有多种,具有代表性是类似于叠氮化合物一些物质,它们具有一般的反应共性,然而也存在相似的弊端。反应膜存在几个问题:第一个问题是失活。随时间的延长,经化学活化的反应膜会因为表面和空气发生反应而失活。膜活化所用
的浓度降低点,这也是个办法。 2)请问有没有合成过配体为FMN的亲和胶? 亲和的填料合成过20来种,你是希望用它来纯化某种脱氢酶吗,我看FMN可偶联的有限,倒是FAD有活泼的氨基好偶联,何况它们几乎是同一个辅酶,你是不是考虑把FAD连上去。当然FMN的结构上也有个仲胺,可以偶联到带长手臂的环氧活化的填料上,而溴化氰活化或别的活化的介质都不大好,因此我觉得这个没有什么问题。 此外如果是以FMN为辅酶的,那我还可以建议你试试蓝色琼脂糖凝胶,因为它的配基的结构和FMN







