相关产品推荐更多 >
万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 询价记录
- 文献和实验
- 技术资料
- 供应商:
青木生物技术(武汉)有限公司
- 库存:
请询
- 英文名:
MycoQsearch Mycoplasma qPCR Detection kit
- 规格:
50 tests/100 tests
| 规格: | 50 tests | 产品价格: | 询价 |
|---|---|---|---|
| 规格: | 100 tests | 产品价格: | 询价 |
- Contains the main contaminants of the cell culture including M. orale, M. hyorhinis, M. arginini, M. fermentans, M. hominis, Acholeplasma laidlawii, allowing all mycoplasma detection of M. pneumoniae, M. salivarium, M. genitalium, M. penentrans and ureaplasma
- The primer binds to the highly conserved 16s rRNA coding region
- DNA of eukaryotes and other bacterial species are not detected
- PCR performance monitoring is possible with the application of internal amplification control (IAC)
- Optimized PCR Premix provided for easy use
- Using the TaqMan probe method, real-time PCR (qPCR) technique is applied
(FAM labeled probe: Mycoplasma, HEX labeld probe: IAC) - Main strand DNA preparation using cell culture solution or commercial DNA extraction kit
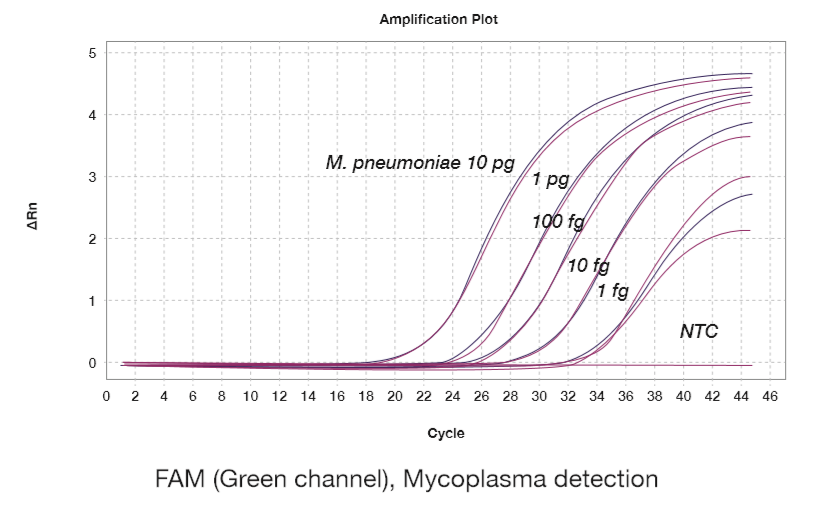
- Sensitivity: 1 ~ 10 fg/reaction
- Result confirmation within 1–2 hr
- Passed the Korea Ministry of Food and Drug Safety (MFDS), European Pharmacopoeia (EP), and Japanese Pharmacopoeia (JP) standards
- UDG system application: Prevention of carry-over contamination

风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
- 作者
- 内容
- 询问日期
 文献和实验
文献和实验Detection of Mycoplasma pneumoniae by Real-Time PCR
-time PCR has emerged as a significant improvement for the rapid diagnosis of this pathogen. The method described herein details the procedure for the detection of M. pneumoniae by real-time PCR (qPCR). The qPCR assay described can be performed with three targets
R&D Systems支原体污染检测新方法(ELISA)专辑
Mycoplasma Detection Kit(Catalog # CUL001B)可以高通量(96T*8种支原体)快速(检测时间也仅需要4.5个小时)有效地检测支原体污染,并且避免出现假阴性和假阳性现象。 支 原体是一类缺乏细胞壁的原核细胞型微生物,大小一般在0.3-0.5um之间,呈高度多形性,有球形、杆形、丝状、分枝状等多种形态。细胞培养(特别是传 代细胞)被支原体污染是个世界性问题。在细胞培养中支原体感染发生率达到63%,支原体感染发生后能改变细胞的DNA,RNA及蛋白表达,又不能
在评估中,故结果仅作为参考和内部品管之一部份。 材料与设备: ATCC mycoplasma detection kit:可作50~100 reactions. 提供1st stage primer mixture与2nd stage primer mixture positive control DNA (A. laidlawii ; M. pirum) PCR reagents : Taq polymerase ( 5 U / ul ) dNTP( dGTP, dCTP, dTTP
 技术资料
技术资料暂无技术资料 索取技术资料