相关产品推荐更多 >

N-(3,5-二甲基苯基)-2-(4-羟基苯基)乙酰胺,131179-77-6,阿拉丁
¥1183.90![(2,3-二氢-[1,4]二氧杂芑并[2,3-b]吡啶-6-基)甲醇,615568-23-5,≥97%,阿拉丁](https://img1.dxycdn.com/p/s14/2026/0106/232/4456575191088158002.jpg!wh200)
(2,3-二氢-[1,4]二氧杂芑并[2,3-b]吡啶-6-基)甲醇,615568-23-5,≥97%,阿拉丁
¥820.90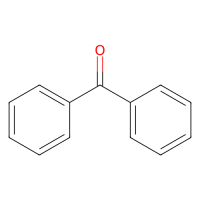
二苯甲酮,119-61-9,适用于合成,阿拉丁
¥94.90
tert-butyl (2R,3S)-3-hydroxy-2-vinyl-piperidine-1-carboxylate,1779500-77-4,≥97%,阿拉丁
¥2242.90![2-(3-氧代-3,4-二氢-2H-苯并[b][1,4]噁嗪-6-基)乙酸甲酯,866038-49-5,≥95%,阿拉丁](https://img1.dxycdn.com/p/s14/2026/0106/048/6319398117197158002.jpg!wh200)
2-(3-氧代-3,4-二氢-2H-苯并[b][1,4]噁嗪-6-基)乙酸甲酯,866038-49-5,≥95%,阿拉丁
¥2717.90
万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 文献和实验
- 技术资料
- 保存条件:
2-8°C储存
- 英文名:
tert-butyl N-(3-hydroxycyclopentyl)-N-methyl-carbamate
- 库存:
期货
- 供应商:
上海阿拉丁生化科技股份有限公司
- CAS号:
1936098-07-5
- 规格:
T678878-5g/T678878-500mg/T678878-25g/T678878-250mg/T678878-1g/T678878-10g/T678878-100mg
| 规格: | T678878-5g | 产品价格: | ¥25584.9 |
|---|---|---|---|
| 规格: | T678878-500mg | 产品价格: | ¥5696.9 |
| 规格: | T678878-25g | 产品价格: | ¥85280.9 |
| 规格: | T678878-250mg | 产品价格: | ¥3424.9 |
| 规格: | T678878-1g | 产品价格: | ¥8528.9 |
| 规格: | T678878-10g | 产品价格: | ¥42640.9 |
| 规格: | T678878-100mg | 产品价格: | ¥2144.9 |
英文名:tert-butyl N-(3-hydroxycyclopentyl)-N-methyl-carbamate
纯度或规格:≥97%
SPU:T678878
CAS:1936098-07-5
存储温度:2-8°C储存
运输条件:低温运输
查看阿拉丁官网此产品相关对应页面:
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 文献和实验
文献和实验Synthesis of Peptides Using Tert-Butyloxycarbonyl (Boc) as the -Amino Protection Group
The use of the tert -butyloxycarbonyl (Boc) as the N α-amino protecting group in peptide synthesis can be advantageous in several cases, such as synthesis of hydrophobic peptides and peptides containing ester and thioester moieties
Enzymes Acting on d-Amino Acid Containing Peptides
aminopeptidase and endopeptidase activities. The enzyme was active toward (d -Phe) n , Boc-(d -Phe) n , (d -Phe) n methyl ester, d -Phe-NH2 , Boc-(d -Phe) n methyl ester, and Boc-(d -Phe) n tert -butyl ester, but not toward (d -Ala) n (n = 2–4), (d
Natural Product Chemistry in Action: The Synthesis of Melatonin Metabolites K1 and K2
melatonin, the kynurenamines N1-acetyl-N2 -formyl-5-methoxykynurenine (K1 ) and N -acetyl-5-methoxykynurenamine (K2 ). The four key synthetic transformations involve (a) conversion of 4-methoxy aniline into the tert -butyl (4-methoxyphenyl)carbamate, (b
 技术资料
技术资料暂无技术资料 索取技术资料