相关产品推荐更多 >
万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 文献和实验
- 技术资料
- 保修期:
12个月
- 现货状态:
现货
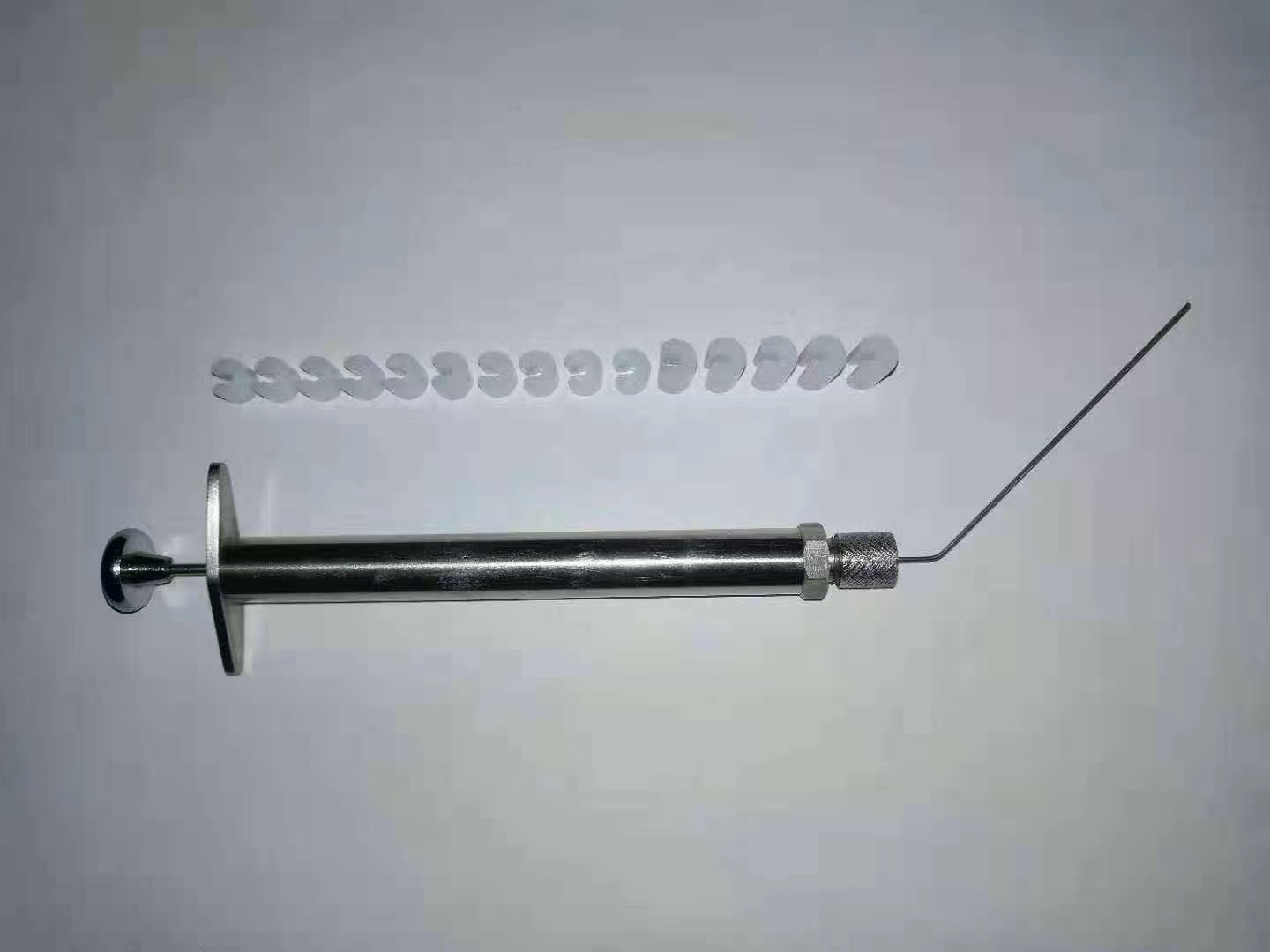
性能特点:
精确定量
较气管内滴入在各肺叶中分布更均匀
直达肺部、易于操作
更安全的提供高浓度
可输送液体、干粉样品
附:北京元森凯德生物技术有限公司2013年成立于北京中关村科技园,是一家专业从事生命科学类实验仪器研制、生产与销售的科技创新型企业。服务毒理学、药理学、免疫学、生物安全、大气污染物、化学物质毒性鉴定、临床前药物开发与安全性评价、呼吸系统、环境与健康等领域。
元森凯德在中国北京、美国宾夕法尼亚均设有技术联络中心,注重仪器的售前、售中、售后沟通,时刻关注行业的新进展动态,客户群体主要有全国各大高校、实验动物科研单位、药物研发机构、第三方CRO及医院中心实验室等。我们将以领先的技术、优质的产品、完善的服务致力于成为业内优秀的实验仪器设备供应厂商。
我们的目标是:服务用户至上,让科研仪器的使用变得更简便和高效。
应用范围:
广泛应用于呼吸系统疾病、毒理学、药理学、吸入免疫、生物安全、大气污染物、化学物质毒性鉴定、药物开发与安全性评价、环境与健康等领域
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 文献和实验
文献和实验Treating Multiorgan Fibrosis
Qiang Long, Zehua Liu, Qianwen Shao, Hongpeng Shi, Shixing Huang, Chenyu Jiang,
Bei Qian, Yiming Zhong, Xiaojun He, Xiaogang Xiang, Yang Yang, Bing Li, Xiaoxiang Yan,
Qiang Zhao,* Xiaoli Wei,* Hélder A. Santos,* and Xiaofeng Ye*
Fibrotic diseases remain a substantial health burden with few therapeutic
approaches. A hallmark of fibrosis is the aberrant activation and accumulation
of myofibroblasts, which is caused by excessive profibrotic cytokines.
Conventional anticytokine therapies fail to undergo clinical trials, as simply
blocking a single or several antifibrotic cytokines cannot abrogate the
profibrotic microenvironment. Here, biomimetic nanoparticles based on
autologous skin fibroblasts are customized as decoys to neutralize multiple
fibroblast-targeted cytokines. By fusing the skin fibroblast membrane onto
poly(lactic-co-glycolic) acid cores, these nanoparticles, termed fibroblast
membrane-camouflaged nanoparticles (FNPs), are shown to effectively
scavenge various profibrotic cytokines, including transforming growth
factor-휷, interleukin (IL)-11, IL-13, and IL-17, thereby modulating the
profibrotic microenvironment. FNPs are sequentially prepared into multiple
formulations for different administration routines. As a proof-of-concept, in
three independent animal models with various organ fibrosis (lung fibrosis,
liver fibrosis, and heart fibrosis), FNPs effectively reduce the accumulation of
myofibroblasts, and the formation of fibrotic tissue, concomitantly restoring
organ function and indicating that FNPs are a potential broad-spectrum
therapy for fibrosis management.
Q. Long, H. Shi, S. Huang, C. Jiang, B. Qian, Y. Zhong, X. He, Q. Zhao,
X. Ye
Department of Cardiovascular Surgery
Ruijin Hospital
Shanghai Jiao Tong University School of Medicine
Shanghai 200025, China
E-mail: zq11607@rjh.com.cn; yxf11612@rjh.com.cn
Z. Liu, H. A. Santos
Department of Biomedical Engineering, W.J. Kolff Institute for
Biomedical Engineering and Materials Science
University Medical Center Groningen/University of Groningen
Ant. Deusinglaan 1, Groningen 9713 AV, The Netherlands
E-mail: h.a.santos@umcg.nl
The ORCID identification number(s) for the author(s) of this article
can be found under https://doi.org/10.1002/advs.202200856
© 2022 The Authors. Advanced Science published by Wiley-VCH GmbH.
This is an open access article under the terms of the Creative Commons
Attribution License, which permits use, distribution and reproduction in
any medium, provided the original work is properly cited.
DOI: 10.1002/advs.202200856
1. Introduction
Fibrosis, or disordered fibrotic tissue formation, is characterized by the abnormal
fibroblast activation that induces excessive extracellular matrix (ECM) remodeling
and primarily accounts for multiple organ
dysfunctions.[1] The pervasive occurrence
of fibrosis in almost all diseases generates
a large healthcare burden worldwide. However, the clinical benefits of antifibrotic therapy through small molecules, such as pirfenidone and nintedanib, are usually offset
by their modest therapeutic efficacy, limited
indications and severe side effects.[2] Therefore, alternative clinical intervention modalities to target fibrosis are urgently needed.
Considering the central role of myofibroblast activation and proliferation in
fibrosis establishment,[3] recent breakthroughs have focused on the ablation
of progressive myofibroblast activation
through autologous cell-based therapy.
For example, autologous chimeric antigen
Z. Liu, H. A. Santos
Drug Research Program
Division of Pharmaceutical Chemistry and Technology
Faculty of Pharmacy
University of Helsinki
Helsinki FI-00014, Finland
Q. Shao, X. Wei
Department of Pharmacology
School of Basic Medical Sciences
Fudan University
Shanghai 200032, China
E-mail: xlwei@fudan.edu.cn
X. Xiang
Department of Infectious Diseases
Ruijin Hospital
Shanghai Jiao Tong University School of Medicine
Shanghai 200025, China
Y. Yang
Department of Thoracic Surgery
Shanghai Pulmonary Hospital
School of Medicine
Tongji University
Shanghai 200000, China
Adv. Sci. 2022, 9, 2200856 2200856 (1 of 14) © 2022 The Authors. Advanced Science published by Wiley-VCH GmbHwww.advancedsciencenews.com www.advancedscience.com
receptor (CAR) T cell therapy to specifically kill myofibroblasts
has achieved unprecedented success in resolving multiorgan
fibrosis.[4] However, the clinical translation of genetically edited
cell therapies may be limited by the exorbitant cost and concomitant immunotoxicity.[5] Therefore, further efforts to develop an
alternative autologous cell-based therapeutic modality with low
cost and satisfactory biocompatibility are also needed.
Instead of directly killing myofibroblasts, specific blockade of
myofibroblast activation represents a promising alternative strategy. Notably, cytokines like transforming growth factor-훽 (TGF-훽)
family proteins, interleukin (IL)-11, IL-13, and IL-17 have been
shown to exert critical roles in mediating fibrosis.[6] Although
some of the anticytokine therapies have been approved by the
FDA with promising results (such as tocilizumab), which brings
a silver lining to the refractory medical issues, Nonetheless, some
of them still suffered from unsatisfied clinical outcomes.[7] This
failure is mainly because: 1) fibrotic disorders involve multiple
cytokines, and simple inhibition of a single or a few types of
cytokines may not be sufficient; and 2) off-target inhibition of
these cytokines may induce severe side effects. Therefore, nextgeneration therapies are expected to use a broad-spectrum and
locally applied anticytokine strategy to target the overall fibrotic
microenvironment.
Here, we developed autologous skin fibroblast-based therapy
to effectively attenuate multiorgan fibrosis. Inactivated autologous skin fibroblasts with intact membrane receptors are prepared in a facile, robust, and economically feasible manner. Endogenous receptors function as decoys to regulate the action of
cytokines, as they can recognize, sequester, and scavenge certain
cytokines but are incapable of triggering signal transduction (Figure 1a). The membrane decoy is supported by a poly(lactic-coglycolic) acid (PLGA)-based nanoparticle cores, termed fibroblast
membrane-camouflaged nanoparticles (FNPs), to enhance stability and facilitate administration. We then examined the competitive binding of multiple profibrotic cytokines with FNPs in
vitro, and the antifibrotic efficacy of FNPs in vivo was confirmed
through three independent animal models with various organ
fibrosis (liver fibrosis, lung fibrosis, and heart fibrosis), which
demonstrates its promising clinical potential (Figure 1b).
2. Results
2.1. Fabrication and Characterization of FNPs
A schematic representation of the fabrication of FNPs is shown
in Figure S1 of the Supporting Information. In brief, mouse
B. Li
Department of Respiratory and Critical Care Medicine
Shanghai Pulmonary Hospital
School of Medicine
Tongji University
Shanghai 200000, China
X. Yan
Department of Cardiovascular Medicine
Ruijin Hospital
Shanghai Jiao Tong University School of Medicine
Shanghai 200025, China
skin fibroblasts were first isolated from the tail tip and expanded
in vitro. Immunofluorescence imaging confirmed the expression of various cytokine receptors, including IL11RA, IL13RA,
IL17RA, and TGF-훽RII, on skin fibroblasts (Figure 1c). Skin fibroblasts were then harvested, homogenized, and subjected to repeated centrifugations to obtain purified membranes. The membranes were coated onto PLGA cores through a sonication process to form FNPs. When visualized with transmission electron
microscopy (TEM), FNPs showed a spherical core–shell structure that indicated unilamellar membrane coatings over the polymeric cores (Figure 1d). Dynamic light scattering (DLS) revealed
that FNPs were ≈20 nm larger than the uncoated PLGA nanoparticles (Figure 1e,f), which is similar to the TEM observations.
Moreover, zeta-potential measurements showed that FNPs possessed a similar surface charge to that of fibroblast vesicles (Figure 1e). FNPs possessed a polymer dispersity index (PDI) of 0.18
(Figure 1g), indicating a homogenous population of nanoparticles, and suggesting acceptability for clinical use.[8] To optimize
the membrane coating efficiency, FNPs were prepared with different membrane protein-to-polymer weight ratios as previously
described.[9] After adjusting with a 1× PBS solution, no apparent
size increase was observed in FNPs prepared with a membrane
protein-to-polymer weight ratio greater than 1:1 (Figure S2, Supporting Information), and this formulation was used for subsequent studies. After their synthesis, FNPs were stored at 4 °C
and demonstrated superior stability within 7 days, as monitored
by DLS (Figure S3, Supporting Information). Moreover, to assure
the physicochemical and biological repeatability of FNPs, a set of
quality assurance standards for their manufacturing was developed as previously described[10] (Table S1, Supporting Information).
Subsequently, we examined the internalization of DiD-labeled
FNPs and DiD-labeled PLGA nanoparticles by primary cardiac fibroblasts (CFBs) and macrophages (RAW 264.7 cells).
FNPs showed significantly decreased uptake by both cell lines
compared to the bare PLGA nanoparticles (Figure 1h,i). However, macrophages showed a higher internalization efficiency of
FNPs than CFBs, indicating the potential clearance of FNPs by
macrophages in vivo. To evaluate the safety of FNPs, PBS or FNPs
(20 mg kg−1) were intravenously injected into healthy mice. After 24 h, compared to mice receiving PBS, mice receiving FNPs
showed no statistically significant differences in immune cell
count (including neutrophils, lymphocytes, and monocytes) or
the levels of proinflammatory cytokines (including IL-6 and TNF-
훼), indicating that FNPs did not provoke immune responses in
vivo (Figure S4, Supporting Information). Next, western blotting showed that the FNPs contained various receptors responsible for cytokine binding, including TGF-훽RII, IL11RA, IL13RA,
and IL17RA (Figure 1j). As a control, we further prepared red
blood cell membrane-camouflaged nanoparticles (RNPs) with a
spherical core–shell structure, size distribution, and PDI similar to those of FNPs (Figure S5, Supporting Information). However, western blotting showed that RNPs had low-to-no expression of the aforementioned cytokine receptors (Figure 1j). We
then tested the binding capacity of FNPs to various profibrotic
cytokines, including IL11, IL13, IL17A, and TGF-훽1, which play
prominent roles in fibrosis progression.[1a] We found that FNPs
but not RNPs, effectively neutralized all four cytokines in a dosedependent manner (Figure 1k). Taken altogether, our findings
Adv. Sci. 2022, 9, 2200856 2200856 (2 of 14) © 2022 The Authors. Advanced Science published by Wiley-VCH GmbHwww.advancedsciencenews.com www.advancedscien
离心机的工作原理是:在超强的离心力作用下,使旋转中的样品逐渐偏离旋转中心,从而达到沉降分离的效果。虽然离心机的原理很简单,但是其适用于大量不同的工业应用和研究实验,离心相关的技术文献非常之多。 实验室常规离心机 台式离心机/贝克曼库尔特Allegra X-5 高速离心机/赛默飞世尔科技Sorvall LYNX 细胞培养离心机套装/海蒂诗 值得注意的是,离心的分离效率与旋转半径成正比,而与转速(指每秒或每分钟的旋转速度,通常用每分钟
(MLV)、鸡白血病病毒(ALV)和猫白血病病毒(FLV)分别能引起大小鼠,鸡和猫白血病。Rous鸡肉瘤病毒可使田鼠、鸡、鸭、鹌鹑、猴、蛇等多种动物发生肉瘤。猫肉瘤肉毒(FSV)可使大鼠、猫、犬和猴发生肉瘤。人类腺病毒能诱发小鼠、田鼠肉瘤和淋巴瘤。(一)诱发性肿瘤动物模型1.肝癌 二乙基亚硝胺(DEN)诱发大白鼠肝癌:取体重250g左右的封闭群大白鼠,雌雄不拘。按性别分笼饲养。除给普通食物外,饲以致癌物,即用 0.25%DEN水溶液灌胃,剂量为10mg/kg,每周一次,其余5天用0.025%DEN水溶
报导,例如小鼠白血病病毒(MLV)、鸡白血病病毒(ALV)和猫白血病病毒(FLV)分别能引起大小鼠,鸡和猫白血病。Rous鸡肉瘤病毒可使田鼠、鸡、鸭、鹌鹑、猴、蛇等多种动物发生肉瘤。猫肉瘤肉毒(FSV)可使大鼠、猫、犬和猴发生肉瘤。人类腺病毒能诱发小鼠、田鼠肉瘤和淋巴瘤。 一、诱发性肿瘤动物模型 1.肝癌 二乙基亚硝胺(DEN)诱发大白鼠肝癌:取体重250g左右的封闭群大白鼠,雌雄不拘。按性别分笼饲养。除给普通食物外,饲以致癌物,即用0.25%DEN水溶液灌胃,剂量为10mg/kg,每周
 技术资料
技术资料暂无技术资料 索取技术资料










