手机验证

InVivoPlus anti-mouse PD-1 (CD279)
InVivoPlus anti-mouse PD-1 (CD279)
随货说明
随货说明
in vivo blocking of PD-1/PD-L signaling, in vitro PD-1 neutralization, Immunohistochemistry (frozen), Flow cytometry, Western blot
随货说明
mouse
体内研究级别
大量
欣博盛生物
无
29F.1A12
4度避光
液体
Rat IgG2a, kappa
mouse PD-1 (CD279)
5 mg
BioXcell InVivoPlus anti-mouse PD-1 29F.1A12单克隆抗体与小鼠PD-1(也称为CD279)反应。PD-1是一种50-55kDa的细胞表面受体,由Pdcd1基因编码,属于Ig超家族的CD28家族。PD-1在CD4和CD8胸腺细胞以及活化的T和B淋巴细胞和髓细胞上瞬时表达,在成功消除抗原后PD-1的表达下降。此外,在B细胞前阶段,Pdcd1 mRNA在发育中的B淋巴细胞中表达。PD-1的结构包括ITIM(基于免疫受体酪氨酸的抑制基序),这表明PD-1负调控TCR信号。PD-1通过结合B7家族的成员PD-L1和PD-L2发出信号。在配体结合后,PD-1信号传导抑制T细胞活化,导致增殖减少,细胞因子产生和T细胞死亡。此外,PD-1敲除动物表现出扩张型心肌病、脾肿大和外周耐受丧失,在小鼠的外周耐受性和预防自身免疫性疾病中发挥关键作用。诱导的PD-L1表达常见于许多肿瘤,包括鳞状细胞癌、结肠癌和乳腺癌。PD-L1过度表达会导致肿瘤细胞对CD8T细胞介导的裂解的抗性增加。在黑素瘤的小鼠模型中,可以通过用阻断PD-L1与其受体PD-1之间相互作用的抗体治疗来暂时抑制肿瘤生长。由于这些原因,目前正在探索抗PD-1介导的免疫疗法作为癌症治疗。与RMP1-14和J43抗体一样,BioXcell InVivoPlus anti-mouse PD-1 29F.1A12抗体在体内可阻断PD-1与其配体的结合。
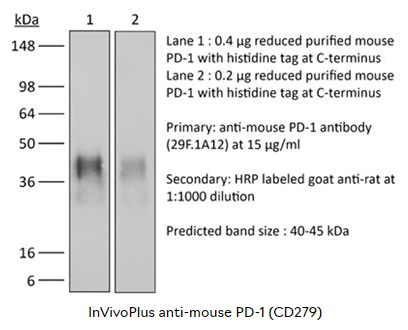
产品详情:
| 产品名称 | InVivoPlus anti-mouse PD-1 (CD279) |
| 产品货号 |
BP0273 |
| 产品规格 |
5/25/50/100mg |
| 反应种属 |
Mouse |
| 克隆号 |
29F.1A12 |
| 同种型 |
Rat IgG2a |
| 免疫原 |
Recombinant PD-1-Ig fusion protein |
| 实验应用 |
in vivo blocking of PD-1/PD-L signaling in vitro PD-1 neutralization Immunohistochemistry (frozen) Immunofluorescence Western blot Flow cytometry |
| 产品形式 |
PBS, pH 7.0,Contains no stabilizers or preservatives |
| 纯度 |
>95%, Determined by SDS-PAGE |
| 聚合 |
<5%, Determined by SEC |
| 无菌处理 |
0.2 µm filtration |
| 纯化方式 |
Protein G |
| RRID |
AB_2687796 |
| 分子量 |
150 kDa |
| 小鼠病原检测 |
Ectromelia/Mousepox Virus: Negative Hantavirus: Negative K Virus: Negative Lactate Dehydrogenase-Elevating Virus: Negative Lymphocytic Choriomeningitis virus: Negative Mouse Adenovirus: Negative Mouse Cytomegalovirus: Negative Mouse Hepatitis Virus: Negative Mouse Minute Virus: Negative Mouse Norovirus: Negative Mouse Parvovirus: Negative Mouse Rotavirus: Negative Mycoplasma Pulmonis: Negative Pneumonia Virus of Mice: Negative Polyoma Virus: Negative Reovirus Screen: Negative Sendai Virus: Negative Theiler’s Murine Encephalomyelitis: Negative |
| 保存条件 |
抗体原液保存在4°C,不能冷冻保存。 |
| 推荐同型对照 |
InVivoPlus rat IgG2a isotype control, anti-trinitrophenol(货号BP0089) |
| 推荐抗体稀释液 |
InVivoPure pH 7.0 Dilution Buffer(货号IP0070) |
为什么选择InVivoPlus抗体?
InVivoPlus级别的产品内毒素含量更低,经过多种实验验证,更适合用于体内实验研究
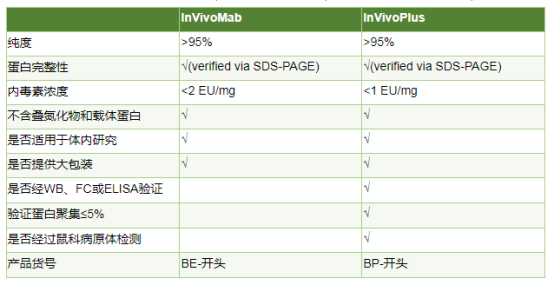
该产品自上市已被多篇SCI文献引用,品质有保证,以下是部分已发表的文献引用:
| 应用 |
文章 |
| 体内PD-1/PD-L信号阻断 (in vivo blocking of PD-1/PD-L signaling) |
1. Wang, W., et al. (2018). 'RIP1 Kinase Drives Macrophage-Mediated Adaptive Immune Tolerance in Pancreatic Cancer' Cancer Cell 34(5): 757-774 e757. 2. Gordon, S. R., et al. (2017). 'PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity' Nature 545(7655): 495-499. 3. Koyama, S., et al. (2016). 'STK11/LKB1 Deficiency Promotes Neutrophil Recruitment and Proinflammatory Cytokine Production to Suppress T-cell Activity in the Lung Tumor Microenvironment' Cancer Res 76(5): 999-1008. |
| 体内PD-1/PD-L信号阻断,流式细胞术 (in vivo blocking of PD-1/PD-L signaling, Flow Cytometry) |
1.Koyama, S., et al. (2016). 'Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints' Nat Commun 7: 10501. |
| 体外PD-1中和 (in vitro PD-1 neutralization) |
1.Park, S. J., et al. (2014). 'Negative role of inducible PD-1 on survival of activated dendritic cells' J Leukoc Biol 95(4): 621-629. |
| 体内PD-1/PD-L信号阻断,体外PD-1中和 (in vivo blocking of PD-1/PD-L signaling, in vitro PD-1 neutralization) |
1.Duraiswamy, J., et al. (2013). 'Dual blockade of PD-1 and CTLA-4 combined with tumor vaccine effectively restores T-cell rejection function in tumors' Cancer Res 73(12): 3591-3603. |
| 流式细胞术 (Flow Cytometry) |
1.Good-Jacobson, K. L., et al. (2012). 'CD80 expression on B cells regulates murine T follicular helper development, germinal center B cell survival, and plasma cell generation' J Immunol 188(9): 4217-4225. |

更多产品详情请咨询 BioXcell 中国授权代理——欣博盛生物
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 文献和实验
文献和实验
Pancreatic ductal adenocarcinoma (PDA) is characterized by immune tolerance and immunotherapeutic resistance. We discovered upregulation of receptor-interacting serine/threonine protein kinase 1 (RIP1) in tumor-associated macrophages (TAMs) in PDA. To study its role in oncogenic progression, we developed a selective small-molecule RIP1 inhibitor with high in vivo exposure. Targeting RIP1 reprogrammed TAMs toward an MHCII(hi)TNFalpha(+)IFNgamma(+) immunogenic phenotype in a STAT1-dependent manner. RIP1 inhibition in TAMs resulted in cytotoxic T cell activation and T helper cell differentiation toward a mixed Th1/Th17 phenotype, leading to tumor immunity in mice and in organotypic models of human PDA. Targeting RIP1 synergized with PD1-and inducible co-stimulator-based immunotherapies. Tumor-promoting effects of RIP1 were independent of its co-association with RIP3. Collectively, our work describes RIP1 as a checkpoint kinase governing tumor immunity.
Programmed cell death protein 1 (PD-1) is an immune checkpoint receptor that is upregulated on activated T cells for the induction of immune tolerance. Tumour cells frequently overexpress the ligand for PD-1, programmed cell death ligand 1 (PD-L1), facilitating their escape from the immune system. Monoclonal antibodies that block the interaction between PD-1 and PD-L1, by binding to either the ligand or receptor, have shown notable clinical efficacy in patients with a variety of cancers, including melanoma, colorectal cancer, non-small-cell lung cancer and Hodgkin’s lymphoma. Although it is well established that PD-1-PD-L1 blockade activates T cells, little is known about the role that this pathway may have in tumour-associated macrophages (TAMs). Here we show that both mouse and human TAMs express PD-1. TAM PD-1 expression increases over time in mouse models of cancer and with increasing disease stage in primary human cancers. TAM PD-1 expression correlates negatively with phagocytic potency against tumour cells, and blockade of PD-1-PD-L1 in vivo increases macrophage phagocytosis, reduces tumour growth and lengthens the survival of mice in mouse models of cancer in a macrophage-dependent fashion. This suggests that PD-1-PD-L1 therapies may also function through a direct effect on macrophages, with substantial implications for the treatment of cancer with these agents.
Despite compelling antitumour activity of antibodies targeting the programmed death 1 (PD-1): programmed death ligand 1 (PD-L1) immune checkpoint in lung cancer, resistance to these therapies has increasingly been observed. In this study, to elucidate mechanisms of adaptive resistance, we analyse the tumour immune microenvironment in the context of anti-PD-1 therapy in two fully immunocompetent mouse models of lung adenocarcinoma. In tumours progressing following response to anti-PD-1 therapy, we observe upregulation of alternative immune checkpoints, notably T-cell immunoglobulin mucin-3 (TIM-3), in PD-1 antibody bound T cells and demonstrate a survival advantage with addition of a TIM-3 blocking antibody following failure of PD-1 blockade. Two patients who developed adaptive resistance to anti-PD-1 treatment also show a similar TIM-3 upregulation in blocking antibody-bound T cells at treatment failure. These data suggest that upregulation of TIM-3 and other immune checkpoints may be targetable biomarkers associated with adaptive resistance to PD-1 blockade.
STK11/LKB1 is among the most commonly inactivated tumor suppressors in non-small cell lung cancer (NSCLC), especially in tumors harboring KRAS mutations. Many oncogenes promote immune escape, undermining the effectiveness of immunotherapies, but it is unclear whether the inactivation of tumor suppressor genes, such as STK11/LKB1, exerts similar effects. In this study, we investigated the consequences of STK11/LKB1 loss on the immune microenvironment in a mouse model of KRAS-driven NSCLC. Genetic ablation of STK11/LKB1 resulted in accumulation of neutrophils with T-cell-suppressive effects, along with a corresponding increase in the expression of T-cell exhaustion markers and tumor-promoting cytokines. The number of tumor-infiltrating lymphocytes was also reduced in LKB1-deficient mouse and human tumors. Furthermore, STK11/LKB1-inactivating mutations were associated with reduced expression of PD-1 ligand PD-L1 in mouse and patient tumors as well as in tumor-derived cell lines. Consistent with these results, PD-1-targeting antibodies were ineffective against Lkb1-deficient tumors. In contrast, treating Lkb1-deficient mice with an IL6-neutralizing antibody or a neutrophil-depleting antibody yielded therapeutic benefits associated with reduced neutrophil accumulation and proinflammatory cytokine expression. Our findings illustrate how tumor suppressor mutations can modulate the immune milieu of the tumor microenvironment, and they offer specific implications for addressing STK11/LKB1-mutated tumors with PD-1-targeting antibody therapies.
PD-1 is a well-established negative regulator of T cell responses by inhibiting proliferation and cytokine production of T cells via interaction with its ligands, B7-H1 (PD-L1) and B7-DC (PD-L2), expressed on non-T cells. Recently, PD-1 was found to be expressed in innate cells, including activated DCs, and plays roles in suppressing production of inflammatory cytokines. In this study, we demonstrate that PD-1 KO DCs exhibited prolonged longevity compared with WT DCs in the dLNs after transfer of DCs into hind footpads. Interestingly, upon LPS stimulation, WT DCs increased the expression of PD-1 and started to undergo apoptosis. DCs, in spleen of LPS-injected PD-1 KO mice, were more resistant to LPS-mediated apoptosis in vivo than WT controls. Moreover, treatment of blocking anti-PD-1 mAb during DC maturation resulted in enhanced DC survival, suggesting that PD-1:PD-L interactions are involved in DC apoptosis. As a result, PD-1-deficient DCs augmented T cell responses in terms of antigen-specific IFN-gamma production and proliferation of CD4 and CD8 T cells to a greater degree than WT DCs. Moreover, PD-1 KO DCs exhibited increased MAPK1 and CD40-CD40L signaling, suggesting a possible mechanism for enhanced DC survival in the absence of PD-1 expression. Taken together, our findings further extend the function of PD-1, which plays an important role in apoptosis of activated DCs and provides important implications for PD-1-mediated immune regulation.


