相关产品推荐更多 >
万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 文献和实验
- 技术资料
- 免疫原:
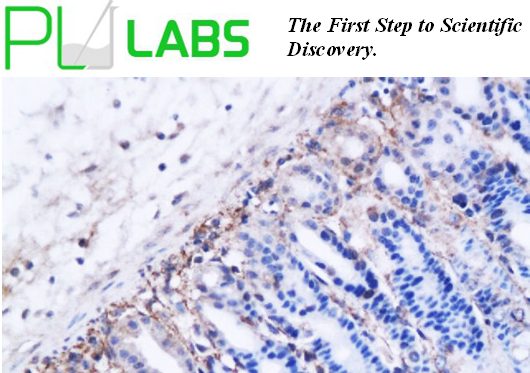
Peptide derived from human TIMP-4.
- 亚型:
Rabbit Polyclonal IgG
- 形态:
液态
- 保存条件:
保存于-20℃
- 克隆性:
多克隆
- 标记物:
无标记(PLlabs可供应生物素、酶、荧光素标记的抗体)
- 适应物种:
Hu, Mo, Rat, Cow, Hor,
- 保质期:
按说明书条件可保存1-2年
- 抗原来源:
见详细说明资料
- 目录编号:
PL0305457
- 级别:
超纯
- 库存:
大量
- 供应商:
华拓生物
- 宿主:
Rabbit
- 应用范围:
WB: 1:100-500 E: 1:500-1000 IP: 1:20-100 IHC: 1:100-500 IF: 1:100-500
- 浓度:
1mg/ml
- 靶点:
TIMP-4
- 抗体英文名:
TIMP-4 Antibody
- 抗体名:
金属蛋白酶组织抑制因子-4抗体
- 规格:
0.1ml

Storage Buffer: PBS, pH 7.4(1%BSA and 0.1% Sodium azide)
Background:
This product for research use only, not for use in humans, therapeutic or diagnostic procedures.


PLLABS坚持提供高质量的产品和专业的技术服务,并提供完善的技术支持和售后服务。
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 文献和实验
文献和实验大鼠基质金属蛋白酶抑制因子9(TIMP-9)酶联免疫分析(ELISA)
大鼠 基质金属蛋白酶抑制因子9(TIMP-9) 酶联免疫分析( ELISA ) 试剂盒使用说明书 本试剂仅供研究使用 目的:本试剂盒用于测定大鼠血清,血浆及相关液体样本中基质金属蛋白酶抑制因子9(TIMP-9) 含量。 实验原理: 本试剂盒应用双抗体夹心法测定标本中大鼠基质金属蛋白酶抑制因子 9(TIMP-9) 水平。用纯化的基质金属蛋白酶抑制因子 9(TIMP-9) 抗体包被微孔板,制成固相抗体,往包被单抗的微孔
人金属蛋白酶组织抑制因子-2(TIMP-2)酶联免疫分析(ELISA)
人金属蛋白酶组织抑制因子-2(TIMP-2) 酶联免疫分析( ELISA ) 试剂盒使用说明书 本试剂仅供研究使用 目的:本试剂盒用于测定人血清,血浆及相关液体样本中金属蛋白酶组织抑制因子-2(TIMP-2) 含量。 实验原理: 本试剂盒应用双抗体夹心法测定标本中 人 金属蛋白酶组织抑制因子 -2(TIMP-2) 水平。用纯化的 人 金属蛋白酶组织抑制因子 -2(TIMP-2) 抗体包被微孔
人金属蛋白酶组织抑制因子-9(TIMP-9)酶联免疫分析(ELISA)
人金属蛋白酶组织抑制因子-9(TIMP-9) 酶联免疫分析( ELISA ) 试剂盒使用说明书 本试剂仅供研究使用 目的:本试剂盒用于测定人血清,血浆及相关液体样本中金属蛋白酶组织抑制因子-9 (TIMP-9) 的 含量。 实验原理: 本试剂盒应用双抗体夹心法测定标本中人金属蛋白酶组织抑制因子 -9 (TIMP-9) 水平。用纯化的人金属蛋白酶组织抑制因子 -9 (TIMP-9) 抗体
 技术资料
技术资料暂无技术资料 索取技术资料









