单抗从头测序(Complete De Novo Sequen
cing of mAb & Other Proteins)- ¥120000
- USA
- 蛋白从头测序
- 2025年07月13日
相关产品推荐更多 >
万千商家帮你免费找货
0 人在求购买到急需产品
- 详细信息
- 文献和实验
- 技术资料
- 提供商:
ProtTech Inc.
| 规格: | 产品价格: | 询价 | |
|---|---|---|---|
| 规格: | 产品价格: | ¥120000.0 |
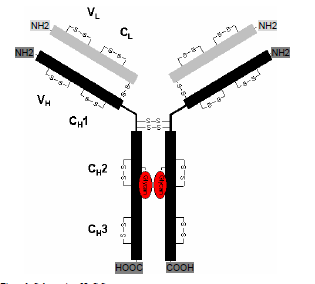
ProtTech®提供单抗全序列从头测序服务,我们建立了抗体从头测序的一系列方法并开发了十多个专有软件,已有完善的数据分析平台,我们能保证最终测序100%的准确。
1:单抗轻重链肽段覆盖率达到100%。
2:首次检测氨基酸准确率达到99.5%以上,活性如果有差异,我们免费复测,找出可能的差异点,保证客户在CHO细胞表达的抗体活性与原来相同。
3:适用于 人源、小鼠源、大鼠源、兔源等的IgG和IgM。
4:样品要求:蛋白纯度95%以上,蛋白量500ug 以上,周期4周。
ProtTech offer the service for the complete de novo sequencing of mAb and other proteins. The service is based on the proprietary protein sequencing technology that we developed over many years. We guarantee a result with a 100% sequencing coverage of the entire mAb molecules, a better than 99.5% accuracy and distinct assignment of Leu/Ile residues.
We also offer an optional confirmation service to confirm the correctness of the variable regions of the sequenced mAb. In this validation process, we express HV and LV region O in E. coli, and use LC/MS/MS peptide mapping to compare each variable region peptide from original and expressed proteins. We gurantee a 100% correctness of the mAb sequence after this confirmation process. Since we use E. coli as the expression host, the confirmation process is often much faster and lower cost than mammalian host expression. Since the sequence confirmation is based on MS and MS/MS of each peptide instead of ELISA assay, the sequence fidelity is much higher.
Procedure:
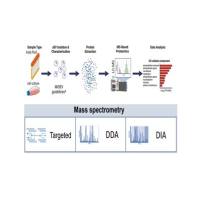
The complete mAb de novo sequencing is often a very complex process, involving more than ten different proteolytic digestions, several steps of chemical derivatizations, NanoLC-ESI-MS/MS data acquisition, bioinformatics analysis with more than a dozen proprietary softwares, the manual spectra assignment, etc. In general the process contains the following steps: 1). Proteolytic digestion and chemical derivatization. 2). LCMS data acquisition. 3). Database search and peptide de novo sequencing. 4). Draft sequencing assembling. 5). Sequence gap filling. 6. Sequence error screening. 7). I/L determination. 8). Sequence validation.
Our mAb de novo sequencing technology can also apply to other proteins. However, changes in the process is often needed based on the nature of the protein.
Service Terms:
Sample requirement: Since more than ten different proteolytic reactions are carried out in the sequencing process, we prefer to have > 1mg mAb protein for each project, although ~500ug mAb sample is acceptable. A better than 90% purity is required. The main concern about the quality of a sample is immunoglobin contamination since there is no easy way to separate contaminating antibodies from the mAb of interest, and there is not easy to determine if a peptide from contaminating peptides or from the mAb of interest. The major source of such a contamination is the serum in cell culture media.
Turn-Around Time:
It often takes five week to sequence one mAb sample.
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 文献和实验
文献和实验De Novo Sequencing Methods in Proteomics
The review describes methods of de novo sequencing of peptides by mass spectrometry. De novo methods utilize computational approaches to deduce the sequence or partial sequence of peptides directly from the experimental MS/MS spectra
网上搜索“抗体从头测序”,搜索结果会展示一长串的资源和服务。关于该技术的描述太多了,有很多描述都大同小异。小编想借此文给大家说说我们为什么需要抗体从头测序以及如何进行抗体从头测序。 抗体从头测序的分类 抗体从头测序是根据抗体样品种类进行区分的。我们通常根据抗体的“克隆性”( clonality)来描述抗体样品。克隆性是指样品中存在的不同抗体序列或遗传系谱的数量。 单克隆:样品中包含具有相同序列的抗体。单克隆性是进行抗体从头测序的前提条件。在极少数情况下,可以对单抗的简单混合物进行蛋白
2019 年 10 月 8 日,美国食品药品监督管理局(FDA)批准了诺华的 Beovu®(brolucizumab)注射剂用于治疗 wet-AMD,这是 FDA 批准的首个兔源单抗药物。Beovu(brolucizumab)的本质是一种人源化单链抗体片段(scFv),是目前达到开发阶段的临床上最先进的人源化单链抗体片段。单链抗体片段体积小、组织渗透性强、可从全身循环里快速清除及其药物释放特性,其在药物开发中备受追捧。单克隆抗体技术起源于 Kohler 等创建的体外鼠源杂交瘤技术。经几十年发展
 技术资料
技术资料暂无技术资料 索取技术资料