手机验证

Bioxcell抗体
Bioxcell antibody
多样
多样
广泛
广泛
欣博盛生物
大量现货
体内研究
BE或BP开头
广泛
一年
小鼠/大鼠
广泛
单克隆
产品说明
产品说明
产品说明
产品说明
1/5/25/50/100mg
BioXCell代理,BioXCell现货库存中心 --欣博盛生物 BioXCell的中和/阻断抗体受到国内外研究者的普遍热爱和青睐,在肿瘤、癌症等方面的研究中广受好评。BioXCell公司位于美国新罕布什尔州,具有25年以上单克隆抗体和重组蛋白的生产及定制经验,可提供高纯度、低内毒素、无防腐剂、适用于体内临床前研究的单克隆抗体。
欣博盛生物是Bioxcell一级代理商,Bioxcell国内唯一库存中心。如需购买Bioxcell公司产品,请联系我们。
 |
高纯度、低内毒素,无防腐剂,适用于体内临床前研究 |
| 超过二十五年单克隆抗体和重组蛋白生产及定制经验 | |
| 性价比高,可提供100mg甚至50g的大包装 | |
| 超过15000篇高质量期刊文献引用 |
肿瘤癌症免疫治疗应用
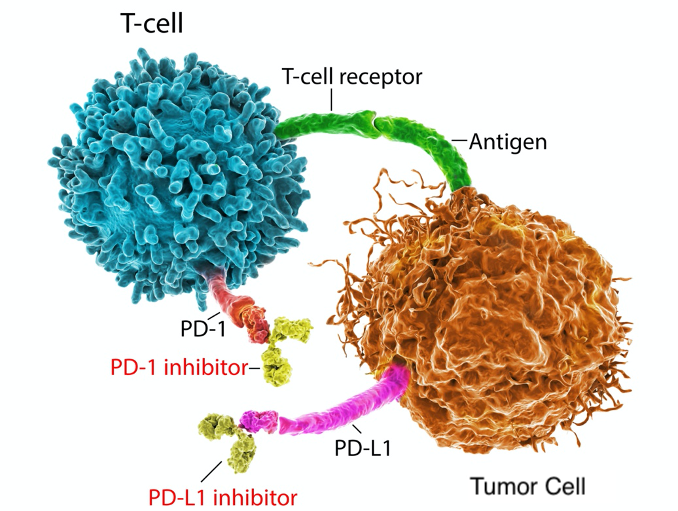
●抑制免疫检查点和其他免疫调节来治疗恶性肿瘤是现在利用免疫系统杀死肿瘤细胞的有力途径。BioXCell提供三种不同克隆号的anti mouse PD-1抗体:RMP1-14、29F.1A12和J43。三种抗体都通过相同的机制发挥作用—它们结合PD-1,并在空间上阻断PD-1与PD-1配体的结合,从而阻断PD-1信号传导。这三种克隆号的抗体都非常适合在小鼠体内模型中阻断PD-1信号传导,并有大量文献支持这一应用。这些抗体之间的差异在于文献报道中的其他应用、Isotype和来源。
●通过用阻断PD-L1和它的受体PD-1之间的相互作用的抗体治疗,肿瘤生长可以暂时被抑制。
●抗PD-1联合抗CTLA-4抗体介导的免疫治疗对黑色素瘤、肾细胞癌和非小细胞肺癌具有显著疗效。
●通过靶向调节免疫反应的途径,如RANK途径,可以将“cold”肿瘤转化为“hot”肿瘤。使用Bio X Cell’s Anti-mouse RANKL (clone IK22/5) antibody,研究人员证明了对RANK通路的抑制将“cold”乳腺肿瘤转化为“hot”肿瘤,变为“hot”的肿瘤可能受益于免疫疗法。
InVivoMab vs. InVivoPlus 如何选择?
| InVivoMab |
InVivoPlus |
|
| 纯度 |
> 95% |
> 95% |
| 蛋白完整性 |
√ (verified via SDS-PAGE) |
ß(verified via SDS-PAGE) |
| 内毒素浓度 |
< 2EU/mg |
< 1EU/mg |
| 不含叠氮化物和载体蛋白 |
√ |
√ |
| 是否适用于体内研究 |
√ |
√ |
| 是否提供大包装 |
√ |
√ |
| 是否经WB, FC或ELISA验证 |
√ |
|
| 经验证蛋白聚集≤ 5% |
√ |
|
| 是否经过鼠科病原体检测 |
√ |
|
| 产品货号 |
BE-开头 |
BP-开头 |
这些指标的抗体最畅销!
肿瘤研究抗体
| 抗原 |
应用 |
InVivoMab 目录号 |
InVivoPlus 目录号 |
| PD-1 (CD279) |
in vivo blocking of PD-1/PD-L signaling, WB |
BE0146 |
BP0146
|
| in vivo blocking of PD-1/PD-L signaling, in vitro PD-1 neutralization |
BE0033-2 |
BP0033-2
|
|
| PD-L1(B7-H1)
|
in vivo PD-L1 blockade, IF, IHC-F, FC, WB
|
BE0101
|
BP0101
|
| CTLA-4 (CD152)
|
in vivo CTLA-4 neutralization, WB
|
BE0164 |
BP0164
|
| in vivo and in vitro CTLA-4 neutralization |
BE0131 |
BP0131 |
|
| CD4 |
in vivo CD4+ T cell depletion, FC
|
BE0003-1 |
BP0003-1
|
| Ly6G |
in vivo neutrophil depletion, in vivo MDSC depletion, IF, IHC-P, IHC-F, FC |
BE0075-1 |
BP0075-1 |
| in vivo depletion of Gr-1+ myeloid cells, FC |
BE0075 |
BP0075 |
|
| CD8α
|
in vivo CD8+T cell depletion
|
BE0061 BE0117 |
BP0061 BP0117 |
| in vivo CD8+ T cell depletion, IF, FC
|
BE0004-1 |
BP0004-1
|
|
| OX40 (CD134)
|
in vivo and in vitro OX40 activation
|
BE0031 |
|
| CSF1R (CD115)
|
in vivo macrophage/monocyte depletion, in vitro CSF-R1 neutralization, FC
|
BE0213 |
|
| CD40 |
in vivoCD40 activation, in vitro B cell stimulation, in vitro DC stimulation
|
BE0016-2 |
BP0016-2
|
| IFNγ
|
in vivo and in vitro IFNγ neutralization, ELISPOT, FC |
BE0055 |
BP0055 |
| GITR
|
in vivo GITR stimulation
|
BE0063 |
|
| CD154 (CD40L)
|
in vivo and in vitro blocking of CD40/CD40L signaling
|
BE0017-1 |
BP0017-1
|
| NK1.1
|
in vivo NK cell depletion, FC
|
BE0036 |
BP0036
|
| TGF-β
|
in vivo TGFβ neutralization, in vitro TGFβ neutralization
|
BE0057 |
BP0057
|
畅销同型对照抗体
| 同型对照 |
InVivoMab 目录号 |
InVivoPlus 目录号 |
| Rat IgG2a Isotype control, FC |
BE0089 |
BP0089 |
| Rat IgG2b Isotype control |
BE0090 |
BP0090 |
| Mouse IgG1 Isotype control |
BE0083 |
BP0083 |
| Rat IgG1 Isotype control |
BE0088 |
BP0088 |
| Mouse IgG2a Isotype control |
BE0085 |
BP0085 |
| Mouse IgG2b Isotype control |
BE0086 |
BP0086 |
| Polyclonal Armenian Hamster IgG |
BE0091 |
BP0091 |
免疫细胞特异性标志物抗体(Immune Cell Specific Depletion Antibodies)
Bioxcell还可提供各种标签抗体
| 产品名称 |
产品编号 |
| ReadyTag anti-c-myc |
RT0263 |
| ReadyTag anti-GST |
RT0264 |
| ReadyTag anti-GFP |
RT0265 |
| ReadyTag anti-6-His |
RT0266 |
| ReadyTag anti-OVA |
RT0267 |
| ReadyTag anti-HA |
RT0268 |
| ReadyTag anti-DDDDK |
RT0269 |
Bioxcell产品清单(部分):
如需获取BIoxcell产品折扣报价,请联系欣博盛生物!
| 品牌 | 产品编号 | 产品名称 | 规格 |
| BioXcell | BE0000-1MG | InVivoMAb anti-mouse CD1d (CD1.1) | 1 mg |
| BioXcell | BE0000-5MG | InVivoMAb anti-mouse CD1d (CD1.1) | 5 mg |
| BioXcell | BE0000-25MG | InVivoMAb anti-mouse CD1d (CD1.1) | 25 mg |
| BioXcell | BE0000-50MG | InVivoMAb anti-mouse CD1d (CD1.1) | 50 mg |
| BioXcell | BE0000-100MG | InVivoMAb anti-mouse CD1d (CD1.1) | 100 mg |
| BioXcell | BE0001-1-1MG | InVivoMAb anti-mouse CD3ε | 1 mg |
| BioXcell | BE0001-1-5MG | InVivoMAb anti-mouse CD3ε | 5 mg |
| BioXcell | BE0001-1-25MG | InVivoMAb anti-mouse CD3ε | 25 mg |
| BioXcell | BE0001-1-50MG | InVivoMAb anti-mouse CD3ε | 50 mg |
| BioXcell | BE0001-1-100MG | InVivoMAb anti-mouse CD3ε | 100 mg |
| BioXcell | BE0001-1FAB-10MG | InVivoMAb anti-mouse CD3ε F(ab')2 fragment | 10 mg |
| BioXcell | BE0001-1FAB-5MG | InVivoMAb anti-mouse CD3ε F(ab')2 fragment | 5 mg |
| BioXcell | BE0001-1FAB-25MG | InVivoMAb anti-mouse CD3ε F(ab')2 fragment | 25 mg |
| BioXcell | BE0001-1FAB-50MG | InVivoMAb anti-mouse CD3ε F(ab')2 fragment | 50 mg |
| BioXcell | BE0001-2-1MG | InVivoMAb anti-human CD3 | 1 mg |
| BioXcell | BE0001-2-5MG | InVivoMAb anti-human CD3 | 5 mg |
| BioXcell | BE0001-2-25MG | InVivoMAb anti-human CD3 | 25 mg |
| BioXcell | BE0001-2-50MG | InVivoMAb anti-human CD3 | 50 mg |
| BioXcell | BE0001-2-100MG | InVivoMAb anti-human CD3 | 100 mg |
| BioXcell | BE0002-1MG | InVivoMAb anti-mouse CD3 | 1 mg |
| BioXcell | BE0002-5MG | InVivoMAb anti-mouse CD3 | 5 mg |
| BioXcell | BE0002-25MG | InVivoMAb anti-mouse CD3 | 25 mg |
| BioXcell | BE0002-50MG | InVivoMAb anti-mouse CD3 | 50 mg |
| BioXcell | BE0002-100MG | InVivoMAb anti-mouse CD3 | 100 mg |
| BioXcell | BE0003-1-1MG | InVivoMAb anti-mouse CD4 | 1 mg |
| BioXcell | BE0003-1-5MG | InVivoMAb anti-mouse CD4 | 5 mg |
| BioXcell | BE0003-1-25MG | InVivoMAb anti-mouse CD4 | 25 mg |
| BioXcell | BE0003-1-50MG | InVivoMAb anti-mouse CD4 | 50 mg |
| BioXcell | BE0003-1-100MG | InVivoMAb anti-mouse CD4 | 100 mg |
| BioXcell | BE0003-2-1MG | InVivoMAb anti-human CD4 | 1 mg |
| BioXcell | BE0003-2-5MG | InVivoMAb anti-human CD4 | 5 mg |
| BioXcell | BE0003-2-25MG | InVivoMAb anti-human CD4 | 25 mg |
| BioXcell | BE0003-2-50MG | InVivoMAb anti-human CD4 | 50 mg |
| BioXcell | BE0003-2-100MG | InVivoMAb anti-human CD4 | 100 mg |
| BioXcell | BE0003-3-1MG | InVivoMAb anti-mouse CD4 | 1 mg |
| BioXcell | BE0003-3-5MG | InVivoMAb anti-mouse CD4 | 5 mg |
| BioXcell | BE0003-3-25MG | InVivoMAb anti-mouse CD4 | 25 mg |
| BioXcell | BE0003-3-50MG | InVivoMAb anti-mouse CD4 | 50 mg |
| BioXcell | BE0003-3-100MG | InVivoMAb anti-mouse CD4 | 100 mg |
| BioXcell | BE0004-1-1MG | InVivoMAb anti-mouse CD8α | 1 mg |
| BioXcell | BE0004-1-5MG | InVivoMAb anti-mouse CD8α | 5 mg |
| BioXcell | BE0004-1-25MG | InVivoMAb anti-mouse CD8α | 25 mg |
| BioXcell | BE0004-1-50MG | InVivoMAb anti-mouse CD8α | 50 mg |
| BioXcell | BE0004-1-100MG | InVivoMAb anti-mouse CD8α | 100 mg |
| BioXcell | BE0004-2-1MG | InVivoMAb anti-human CD8α | 1 mg |
| BioXcell | BE0004-2-5MG | InVivoMAb anti-human CD8α | 5 mg |
| BioXcell | BE0004-2-25MG | InVivoMAb anti-human CD8α | 25 mg |
| BioXcell | BE0004-2-50MG | InVivoMAb anti-human CD8α | 50 mg |
| BioXcell | BE0004-2-100MG | InVivoMAb anti-human CD8α | 100 mg |
| BioXcell | BE0005-1MG | InVivoMAb anti-human LFA-1α (CD11a) | 1 mg |
| BioXcell | BE0005-5MG | InVivoMAb anti-human LFA-1α (CD11a) | 5 mg |
| BioXcell | BE0005-25MG | InVivoMAb anti-human LFA-1α (CD11a) | 25 mg |
| BioXcell | BE0005-50MG | InVivoMAb anti-human LFA-1α (CD11a) | 50 mg |
| BioXcell | BE0005-100MG | InVivoMAb anti-human LFA-1α (CD11a) | 100 mg |
| BioXcell | BE0005-1-1MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 1 mg |
| BioXcell | BE0005-1-5MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 5 mg |
| BioXcell | BE0005-1-25MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 25 mg |
| BioXcell | BE0005-1-50MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 50 mg |
| BioXcell | BE0005-1-100MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 100 mg |
| BioXcell | BE0006-1MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 1 mg |
| BioXcell | BE0006-5MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 5 mg |
| BioXcell | BE0006-25MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 25 mg |
| BioXcell | BE0006-50MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 50 mg |
| BioXcell | BE0006-100MG | InVivoMAb anti-mouse LFA-1α (CD11a) | 100 mg |
| BioXcell | BE0007-1MG | InVivoMAb anti-mouse/human CD11b | 1 mg |
| BioXcell | BE0007-5MG | InVivoMAb anti-mouse/human CD11b | 5 mg |
| BioXcell | BE0007-25MG | InVivoMAb anti-mouse/human CD11b | 25 mg |
| BioXcell | BE0007-50MG | InVivoMAb anti-mouse/human CD11b | 50 mg |
| BioXcell | BE0007-100MG | InVivoMAb anti-mouse/human CD11b | 100 mg |
| BioXcell | BE0009-1MG | InVivoMAb anti-mouse CD18 | 1 mg |
| BioXcell | BE0009-5MG | InVivoMAb anti-mouse CD18 | 5 mg |
| BioXcell | BE0009-25MG | InVivoMAb anti-mouse CD18 | 25 mg |
| BioXcell | BE0009-50MG | InVivoMAb anti-mouse CD18 | 50 mg |
| BioXcell | BE0009-100MG | InVivoMAb anti-mouse CD18 | 100 mg |
| BioXcell | BE0011-1MG | InVivoMAb anti-mouse CD22 | 1 mg |
| BioXcell | BE0011-5MG | InVivoMAb anti-mouse CD22 | 5 mg |
| BioXcell | BE0011-25MG | InVivoMAb anti-mouse CD22 | 25 mg |
| BioXcell | BE0011-50MG | InVivoMAb anti-mouse CD22 | 50 mg |
| BioXcell | BE0011-100MG | InVivoMAb anti-mouse CD22 | 100 mg |
| BioXcell | BE0012-1MG | InVivoMAb anti-mouse CD25 (IL-2Rα) | 1 mg |
| BioXcell | BE0012-5MG | InVivoMAb anti-mouse CD25 (IL-2Rα) | 5 mg |
| BioXcell | BE0012-25MG | InVivoMAb anti-mouse CD25 (IL-2Rα) | 25 mg |
| BioXcell | BE0012-50MG | InVivoMAb anti-mouse CD25 (IL-2Rα) | 50 mg |
| BioXcell | BE0012-100MG | InVivoMAb anti-mouse CD25 (IL-2Rα) | 100 mg |
| BioXcell | BE0014-1MG | InVivoMAb anti-human CD25 (IL-2Rα) | 1 mg |
| BioXcell | BE0014-5MG | InVivoMAb anti-human CD25 (IL-2Rα) | 5 mg |
| BioXcell | BE0014-25MG | InVivoMAb anti-human CD25 (IL-2Rα) | 25 mg |
| BioXcell | BE0014-50MG | InVivoMAb anti-human CD25 (IL-2Rα) | 50 mg |
| BioXcell | BE0014-100MG | InVivoMAb anti-human CD25 (IL-2Rα) | 100 mg |
| BioXcell | BE0015-1-1MG | InVivoMAb anti-mouse CD28 | 1 mg |
| BioXcell | BE0015-1-5MG | InVivoMAb anti-mouse CD28 | 5 mg |
| BioXcell | BE0015-1-25MG | InVivoMAb anti-mouse CD28 | 25 mg |
| BioXcell | BE0015-1-50MG | InVivoMAb anti-mouse CD28 | 50 mg |
| BioXcell | BE0015-1-100MG | InVivoMAb anti-mouse CD28 | 100 mg |
| BioXcell | BE0015-5-1MG | InVivoMAb anti-mouse CD28 | 1 mg |
| BioXcell | BE0015-5-5MG | InVivoMAb anti-mouse CD28 | 5 mg |
| BioXcell | BE0015-5-25MG | InVivoMAb anti-mouse CD28 | 25 mg |
| BioXcell | BE0015-5-50MG | InVivoMAb anti-mouse CD28 | 50 mg |
| BioXcell | BE0015-5-100MG | InVivoMAb anti-mouse CD28 | 100 mg |
| BioXcell | BE0016-2-1MG | InVivoMAb anti-mouse CD40 | 1 mg |
| BioXcell | BE0016-2-5MG | InVivoMAb anti-mouse CD40 | 5 mg |
| BioXcell | BE0016-2-25MG | InVivoMAb anti-mouse CD40 | 25 mg |
| BioXcell | BE0016-2-50MG | InVivoMAb anti-mouse CD40 | 50 mg |
| BioXcell | BE0016-2-100MG | InVivoMAb anti-mouse CD40 | 100 mg |
| BioXcell | BE0017-1-1MG | InVivoMAb anti-mouse CD40L (CD154) | 1 mg |
| BioXcell | BE0017-1-5MG | InVivoMAb anti-mouse CD40L (CD154) | 5 mg |
| BioXcell | BE0017-1-25MG | InVivoMAb anti-mouse CD40L (CD154) | 25 mg |
| BioXcell | BE0017-1-50MG | InVivoMAb anti-mouse CD40L (CD154) | 50 mg |
| BioXcell | BE0017-1-100MG | InVivoMAb anti-mouse CD40L (CD154) | 100 mg |
| BioXcell | BE0019-1MG | InVivoMAb anti-mouse CD45RB | 1 mg |
| BioXcell | BE0019-5MG | InVivoMAb anti-mouse CD45RB | 5 mg |
| BioXcell | BE0019-25MG | InVivoMAb anti-mouse CD45RB | 25 mg |
| BioXcell | BE0019-50MG | InVivoMAb anti-mouse CD45RB | 50 mg |
| BioXcell | BE0019-100MG | InVivoMAb anti-mouse CD45RB | 100 mg |
| BioXcell | BE0019-1-1MG | InVivoMAb anti-human CD47 | 1 mg |
| BioXcell | BE0019-1-5MG | InVivoMAb anti-human CD47 | 5 mg |
| BioXcell | BE0019-1-25MG | InVivoMAb anti-human CD47 | 25 mg |
| BioXcell | BE0019-1-50MG | InVivoMAb anti-human CD47 | 50 mg |
| BioXcell | BE0019-1-100MG | InVivoMAb anti-human CD47 | 100 mg |
| BioXcell | BE0020-1-1MG | InVivoMAb anti-mouse CD54 (ICAM-1) | 1 mg |
| BioXcell | BE0020-1-5MG | InVivoMAb anti-mouse CD54 (ICAM-1) | 5 mg |
| BioXcell | BE0020-1-25MG | InVivoMAb anti-mouse CD54 (ICAM-1) | 25 mg |
| BioXcell | BE0020-1-50MG | InVivoMAb anti-mouse CD54 (ICAM-1) | 50 mg |
| BioXcell | BE0020-1-100MG | InVivoMAb anti-mouse CD54 (ICAM-1) | 100 mg |
| BioXcell | BE0020-2-1MG | InVivoMAb anti-human CD54 (ICAM-1) | 1 mg |
| BioXcell | BE0020-2-5MG | InVivoMAb anti-human CD54 (ICAM-1) | 5 mg |
| BioXcell | BE0020-2-25MG | InVivoMAb anti-human CD54 (ICAM-1) | 25 mg |
| BioXcell | BE0020-2-50MG | InVivoMAb anti-human CD54 (ICAM-1) | 50 mg |
| BioXcell | BE0020-2-100MG | InVivoMAb anti-human CD54 (ICAM-1) | 100 mg |
| BioXcell | BE0021-1MG | InVivoMAb anti-mouse L-Selectin (CD62L) | 1 mg |
| BioXcell | BE0021-5MG | InVivoMAb anti-mouse L-Selectin (CD62L) | 5 mg |
| BioXcell | BE0021-25MG | InVivoMAb anti-mouse L-Selectin (CD62L) | 25 mg |
| BioXcell | BE0021-50MG | InVivoMAb anti-mouse L-Selectin (CD62L) | 50 mg |
| BioXcell | BE0021-100MG | InVivoMAb anti-mouse L-Selectin (CD62L) | 100 mg |
| BioXcell |
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 询价记录
询价记录
 文献和实验
文献和实验
CD4(+) T cell differentiation into multiple T helper (Th) cell lineages is critical for optimal adaptive immune responses. This report identifies an intrinsic mechanism by which programmed death-1 receptor (PD-1) signaling imparted regulatory phenotype to Foxp3(+) Th1 cells (denoted as Tbet(+)iTregPDL1 cells) and inducible regulatory T (iTreg) cells. Tbet(+)iTregPDL1 cells prevented inflammation in murine models of experimental colitis and experimental graft versus host disease (GvHD). Programmed death ligand-1 (PDL-1) binding to PD-1 imparted regulatory function to Tbet(+)iTregPDL1 cells and iTreg cells by specifically downregulating endo-lysosomal protease asparaginyl endopeptidase (AEP). AEP regulated Foxp3 stability and blocking AEP imparted regulatory function in Tbet(+)iTreg cells. Also, Aep(-/-) iTreg cells significantly inhibited GvHD and maintained Foxp3 expression. PD-1-mediated Foxp3 maintenance in Tbet(+) Th1 cells occurred both in tumor infiltrating lymphocytes (TILs) and during chronic viral infection. Collectively, this report has identified an intrinsic function for PD-1 in maintaining Foxp3 through proteolytic pathway.
T follicular regulatory (Tfr) cells are a subset of Foxp3(+) regulatory T (Treg) cells that form in response to immunization or infection, which localize to the germinal centre where they control the magnitude of the response. Despite an increased interest in the role of Tfr cells in humoral immunity, many fundamental aspects of their biology remain unknown, including whether they recognize self- or foreign antigen. Here we show that Tfr cells can be specific for the immunizing antigen, irrespective of whether it is a self- or foreign antigen. We show that, in addition to developing from thymic derived Treg cells, Tfr cells can also arise from Foxp3(-) precursors in a PD-L1-dependent manner, if the adjuvant used is one that supports T-cell plasticity. These findings have important implications for Tfr cell biology and for improving vaccine efficacy by formulating vaccines that modify the Tfr:Tfh cell ratio.
Despite successes, thus far, a significant proportion of the patients treated with anti-PD1 antibodies have failed to respond. We use mouse tumor models of anti-PD1 sensitivity and resistance and flow cytometry to assess tumor-infiltrating immune cells immediately after therapy. We demonstrate that the expression levels of T-cell PD1 (PD1(lo)), myeloid, and T-cell PDL1 (PDL1(hi)) in the tumor microenvironment inversely correlate and dictate the efficacy of anti-PD1 mAb and function of intratumor CD8(+) T cells. In sensitive tumors, we reveal a threshold for PD1 downregulation on tumor-infiltrating CD8(+) T cells below which the release of adaptive immune resistance is achieved. In contrast, PD1(hi) T cells in resistant tumors fail to be rescued by anti-PD1 therapy and remain dysfunctional unless intratumor PDL1(lo) immune cells are targeted. Intratumor Tregs are partly responsible for the development of anti-PD1-resistant tumors and PD1(hi) CD8(+) T cells. Our analyses provide a framework to interrogate intratumor CD8(+) T-cell PD1 and immune PDL1 levels and response in human cancer. Cancer Res; 75(18); 3800-11. (c)2015 AACR.
Acute kidney injury (AKI) is a common problem in hospitalized patients that enhances morbidity and mortality and promotes the development of chronic and end-stage renal disease. Ischemia reperfusion injury (IRI) is one of the major causes of AKI and is characterized by uncontrolled renal inflammation and tubular epithelial cell death. Our recent studies demonstrated that regulatory T cells (Tregs) protect the kidney from ischemia reperfusion-induced inflammation and injury. Blockade of programmed death-1 (PD-1) on the surface of Tregs, prior to adoptive transfer, negates their ability to protect against ischemic kidney injury. The present study was designed to investigate the role of the known PD-1 ligands, PD-L1 and PD-L2, in kidney IRI. Administration of PD-L1 or PD-L2 blocking Abs prior to mild or moderate kidney IRI significantly exacerbated the loss of renal function, renal inflammation, and acute tubular necrosis compared with mice receiving isotype control Abs. Interestingly, blockade of both PD-1 ligands resulted in worse injury, dysfunction, and inflammation than did blocking either ligand alone. Genetic deficiency of either PD-1 ligand also exacerbated kidney dysfunction and acute tubular necrosis after subthreshold ischemia. Bone marrow chimeric studies revealed that PD-L1 expressed on non-bone marrow-derived cells is critical for this resistance to IRI. Finally, blockade of either PD-1 ligand negated the protective ability of adoptively transferred Tregs in IRI. These findings suggest that PD-L1 and PD-L2 are nonredundant aspects of the natural protective response to ischemic injury and may be novel therapeutic targets for AKI.
CD8(+) T cells activated without CD4(+) T-cell help are impaired in memory expansion. To understand the underlying cellular mechanism, here we track the dynamics of helper-deficient CD8(+) T-cell response to a minor histocompatibility antigen by phenotypic and in vivo imaging analyses. Helper-deficient CD8(+) T cells show reduced burst expansion, rapid peripheral egress, delayed antigen clearance and continuous activation, and are eventually exhausted. Contrary to the general consensus that CD4 help encodes memory programmes in CD8(+) T cells and helper-deficient CD8(+) T cells are abortive, these cells can differentiate into effectors and memory precursors. Importantly, accelerating antigen clearance or simply increasing the burst effector size enables generation of memory cells by CD8(+) T cells, regardless of CD4 help. These results suggest that the memory programme is CD8(+) T-cell-intrinsic, and provide insight into the role of CD4 help in CD8(+) T-cell responses.
The differentiation and protective capacity of Plasmodium-specific T cells are regulated by both positive and negative signals during malaria, but the molecular and cellular details remain poorly defined. Here we show that malaria patients and Plasmodium-infected rodents exhibit atypical expression of the co-stimulatory receptor OX40 on CD4 T cells and that therapeutic enhancement of OX40 signaling enhances helper CD4 T cell activity, humoral immunity, and parasite clearance in rodents. However, these beneficial effects of OX40 signaling are abrogated following coordinate blockade of PD-1 co-inhibitory pathways, which are also upregulated during malaria and associated with elevated parasitemia. Co-administration of biologics blocking PD-1 and promoting OX40 signaling induces excessive interferon-gamma that directly limits helper T cell-mediated support of humoral immunity and decreases parasite control. Our results show that targeting OX40 can enhance Plasmodium control and that crosstalk between co-inhibitory and co-stimulatory pathways in pathogen-specific CD4 T cells can impact pathogen clearance.
The coinhibitory receptor programmed death-1 (PD-1) maintains immune homeostasis by negatively regulating T cell function and survival. Blockade of PD-1 increases the severity of graft-versus-host disease (GVHD), but the interplay between PD-1 inhibition and T cell metabolism is not well studied. We found that both murine and human alloreactive T cells concomitantly upregulated PD-1 expression and increased levels of reactive oxygen species (ROS) following allogeneic bone marrow transplantation. This PD-1(Hi)ROS(Hi) phenotype was specific to alloreactive T cells and was not observed in syngeneic T cells during homeostatic proliferation. Blockade of PD-1 signaling decreased both mitochondrial H2O2 and total cellular ROS levels, and PD-1-driven increases in ROS were dependent upon the oxidation of fatty acids, because treatment with etomoxir nullified changes in ROS levels following PD-1 blockade. Downstream of PD-1, elevated ROS levels impaired T cell survival in a process reversed by antioxidants. Furthermore, PD-1-driven changes in ROS were fundamental to establishing a cell’s susceptibility to subsequent metabolic inhibition, because blockade of PD-1 decreased the efficacy of later F1F0-ATP synthase modulation. These data indicate that PD-1 facilitates apoptosis in alloreactive T cells by increasing ROS in a process dependent upon the oxidation of fat. In addition, blockade of PD-1 undermines the potential for subsequent metabolic inhibition, an important consideration given the increasing use of anti-PD-1 therapies in the clinic.
Immune checkpoint inhibitors result in impressive clinical responses, but optimal results will require combination with each other and other therapies. This raises fundamental questions about mechanisms of non-redundancy and resistance. Here we report major tumour regressions in a subset of patients with metastatic melanoma treated with an anti-CTLA4 antibody (anti-CTLA4) and radiation, and reproduced this effect in mouse models. Although combined treatment improved responses in irradiated and unirradiated tumours, resistance was common. Unbiased analyses of mice revealed that resistance was due to upregulation of PD-L1 on melanoma cells and associated with T-cell exhaustion. Accordingly, optimal response in melanoma and other cancer types requires radiation, anti-CTLA4 and anti-PD-L1/PD-1. Anti-CTLA4 predominantly inhibits T-regulatory cells (Treg cells), thereby increasing the CD8 T-cell to Treg (CD8/Treg) ratio. Radiation enhances the diversity of the T-cell receptor (TCR) repertoire of intratumoral T cells. Together, anti-CTLA4 promotes expansion of T cells, while radiation shapes the TCR repertoire of the expanded peripheral clones. Addition of PD-L1 blockade reverses T-cell exhaustion to mitigate depression in the CD8/Treg ratio and further encourages oligoclonal T-cell expansion. Similarly to results from mice, patients on our clinical trial with melanoma showing high PD-L1 did not respond to radiation plus anti-CTLA4, demonstrated persistent T-cell exhaustion, and rapidly progressed. Thus, PD-L1 on melanoma cells allows tumours to escape anti-CTLA4-based therapy, and the combination of radiation, anti-CTLA4 and anti-PD-L1 promotes response and immunity through distinct mechanisms.
One question that continues to challenge influenza A research is why some strains of virus are so devastating compared to their more mild counterparts. We approached this question from an immunological perspective, investigating the CD8(+) T cell response in a mouse model system comparing high- and low-pathological influenza virus infections. Our findings reveal that the early (day 0 to 5) viral titer was not the determining factor in the outcome of disease. Instead, increased numbers of antigen-specific CD8(+) T cells and elevated effector function on a per-cell basis were found in the low-pathological infection and correlated with reduced illness and later-time-point (day 6 to 10) viral titer. High-pathological infection was associated with increased PD-1 expression on influenza virus-specific CD8(+) T cells, and blockade of PD-L1 in vivo led to reduced virus titers and increased CD8(+) T cell numbers in high- but not low-pathological infection, though T cell functionality was not restored. These data show that high-pathological acute influenza virus infection is associated with a dysregulated CD8(+) T cell response, which is likely caused by the more highly inflamed airway microenvironment during the early days of infection. Therapeutic approaches specifically aimed at modulating innate airway inflammation may therefore promote efficient CD8(+) T cell activity. We show that during a severe influenza virus infection, one type of immune cell, the CD8 T cell, is less abundant and less functional than in a more mild infection. This dysregulated T cell phenotype correlates with a lower rate of virus clearance in the severe infection and is partially regulated by the expression of a suppressive coreceptor called PD-1. Treatment with an antibody that blocks PD-1 improves T cell functionality and increases virus clearance.
Antibodies (Abs) that preferentially target oncogenic receptors have been increasingly used for cancer therapy, but tumors often acquire intrinsic Ab resistance after prolonged and costly treatment. Herein we armed the Ab with IFNbeta and observed that it is more potent than the first generation of Ab for controlling Ab-resistant tumors. This strategy controls Ab resistance by rebridging suppressed innate and adaptive immunity in the tumor microenvironment. Mechanistically, Ab-IFNbeta therapy primarily and directly targets intratumoral dendritic cells, which reactivate CTL by increasing antigen cross-presentation within the tumor microenvironment. Additionally, blocking PD-L1, which is induced by Ab-IFNbeta treatment, overcomes treatment-acquired resistance and completely eradicates established tumors. This study establishes a next-generation Ab-based immunotherapy that targets and eradicates established Ab-resistant tumors.
 技术资料
技术资料
需要更多技术资料 索取更多技术资料
BioXCell——CD Marker抗体.pdf 附 (下载 0 次)


