手机验证

大量
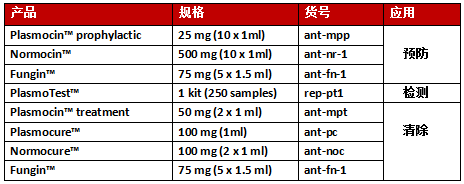
Mycoplasma Removal Agent
研卉生物
低温
25mg
抗微生物污染试剂
 Plasmocin™ can also be used as a routine addition in liquid media to prevent mycoplasma and more generally bacterial contamination in small and large animal cell cultures.
Plasmocin™ can also be used as a routine addition in liquid media to prevent mycoplasma and more generally bacterial contamination in small and large animal cell cultures.
Plasmocin™ is a well-established antimycoplasma reagent. It contains two bactericidal components strongly active against mycoplasmas that allow their elimination in only 2 weeks.
The first component acts on the protein synthesis machinery while the second acts on the DNA replication. These two specific and separate targets are found only in mycoplasmas and many other bacteria and are completely absent in eukaryotic cells.
Warning: InvivoGen's anti-mycoplasma products are suitable for research purposes only, and not for human or animal care.
| Product | Supplier | Treatment | Ease of use | Efficacy | Cytotoxicity | Resistance |
|---|---|---|---|---|---|---|
| BM-Cyclin | Roche | 3 weeks | - | +++ | + | +/- |
| Ciprobay | Bayer | 12 to 20 days | + | ++ | +/- | + |
| MRA | ICN | 1 to 2 weeks | + | ++ | +/- | + |
| Plasmocin | InvivoGen | 2 weeks | + | +++ | +/- | - |
Antibiotics commonly used in cell culture are inactive on mycoplasma (e.g. penicillins and streptomycin). Three classes of antibiotics have been shown to kill mycoplasma at relatively low concentrations: tetracyclines, macrolides and quinolones. Tetracyclines and macrolides block the protein synthesis by interfering with ribosome translation, while quinolones inhibit the replication of bacterial DNA.
Several antibiotics are commercially available for the removal of mycoplasma: BM-cyclin (Roche) contains a macrolide and a tetracycline, Ciprobay (Bayer, available only with a prescription) and MRA (ICN) are both quinolones. Plasmocin™ is the only antimycoplasma reagent that combines a macrolide and a quinolone. Unlike BM-Cyclin that requires the sequential and cyclic use of 2 antibiotics, Plasmocin™ is ready-to-use and can be added to the culture medium directly. Furthermore, the 2 antibiotics in Plasmocin™ act on separate targets blocking protein synthesis and DNA replication, whereas the 2 antibiotics in BM-Cyclin are both inhibitors of protein synthesis. Therefore, Plasmocin™ is more effective in removing mycoplasma and prevents the appearance of resistant strains. In contrast to other anti-mycoplasma compounds, Plasmocin™ is active on both free mycoplasma as well as intracellular forms. This advantage is conferred by one component of Plasmocin™ which is actively transported into mammalian cells. It ensures that following treatment with Plasmocin™ a cell culture is not reinfected by mycoplasma released from intracellular compartments of infected cells. To date, no consistent and permanent alterations that affect the eukaryotic cells during and after the treatment have been detected[1].
1. Uphoff CC, Drexler HG., 2005. Eradication of mycoplasma contaminations. Methods Mol Biol. 290:25-34.
2. Somasundaram C. et al., 1992. Use of ciprofloxacin and BM-Cyclin in mycoplasma decontamination.In Vitro Cell Dev Biol. 28A(11-12):708-10
3. Drexler HG. et al., 1994. Treatment of mycoplasma contamination in a large panel of cell cultures. In vitro Cell Dev Biol Anim. 30A(5):344-7
 Plasmocin™可以用来消除那些被支原体和某些无细胞壁的细菌感染的细胞系。
Plasmocin™可以用来消除那些被支原体和某些无细胞壁的细菌感染的细胞系。
与其它抗支原体试剂相比,Plasmocin™可同时作用于细胞外与细胞内的支原体。这一优点得益于Plasmocin™其中的一个组份可被转运到哺乳动物细胞内,从而确保Plasmocin™处理后的细胞不会因为细胞释放出来的支原体而再此污染。
目前在所有检测的动物细胞中,即使提高至五倍工作浓度,细胞代谢并没有出现明显变化。
多组独立实验结果显示,按说明书处理后的细胞培养液中不含抗性支原体突变株,证明试剂不会引起支原体抗性株。
Plasmocin™也可作用于低浓度的青霉素,链霉素抗性革兰氏阳性和革兰氏阴性菌。
包括胚胎干细胞,杂交瘤细胞和反转录病毒包装细胞在内的多类细胞的支原体污染,均可被Plasmocin™清除。
抗支原体污染常用试剂比较[1-3]
风险提示:丁香通仅作为第三方平台,为商家信息发布提供平台空间。用户咨询产品时请注意保护个人信息及财产安全,合理判断,谨慎选购商品,商家和用户对交易行为负责。对于医疗器械类产品,请先查证核实企业经营资质和医疗器械产品注册证情况。
 询价记录
询价记录
 文献和实验
文献和实验
 技术资料
技术资料
需要更多技术资料 索取更多技术资料
支原体预防 附 (下载 8 次)


